
Defective Medtronic Heart Pump Is Taking Lives? Medtronic Life-Threatening Heart Devices. The Medtronic heart pump is an HVAD System which takes over for a failing heart. Basically, the system helps to support individuals with heart diseases while they wait for a heart transplant. Flaws, however, surfaced with the heart device that the U.S. Food and Drug Administration […]
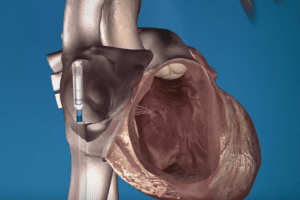
Defective Medtronic Heart Pump Is Taking Lives? Medtronic Life-Threatening Heart Devices. The Medtronic heart pump is an HVAD System which takes over for a failing heart.
Basically, the system helps to support individuals with heart diseases while they wait for a heart transplant. Flaws, however, surfaced with the heart device that the U.S. Food and Drug Administration (FDA) found to be life-threatening.
One patient may have died as a result of a flaw where moisture may penetrate the mechanical pump through loose power or data connectors, said Chris Garland, a spokesman for Medtronic. Medtronic acquired the medical device with its purchase last month of HeartWare International as part of a $1 billion deal, according to Bloomberg News.
In a statement issued several days ago, Medtronic said that doctors and patients were initially notified about the problem in May and June, with instructions to carefully monitor the HVAD System for loose connections. Since then, the FDA has classified the problem a Class 1 recall, which means the issue is potentially life-threatening.
These recall resignations are meant to classify an issue’s severity, and does not mean necessarily that the devices should be removed from patients and replaced, reports Bloomberg News.
Medtronic announced that nearly all 8,799 patients who might be affected by this issue have been notified, and 308 controllers have been replaced. Last month, the company recalled the pump kits that were in hospital inventories as they were susceptible to electrical faults and connection failures from exposure to moisture. Medtronic has recovered 323 of the 350 at-risk kits.
According to Investopedia.com, Medtronic has the largest market share in the cardio device market. They generated over $9.3 billion in cardio-device related sales in 2014.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


