
MRSA Skin Infections Are Taking E.R. Doctors Back To The Pre-Penicillin Days. Methicillin-resistant bacteria are taking emergency room doctors back to the pre-penicillin days, when skin infections were lanced and drained. Methicillin-resistant Staphylococcus aureus (MRSA) infection has become the most common cause of skin and soft tissue infections seen in metropolitan emergency departments, and half-century-old therapeutic […]
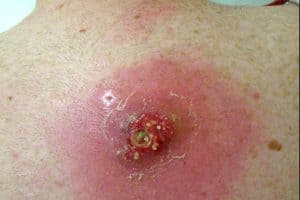
MRSA Skin Infections Are Taking E.R. Doctors Back To The Pre-Penicillin Days. Methicillin-resistant bacteria are taking emergency room doctors back to the pre-penicillin days, when skin infections were lanced and drained.
Methicillin-resistant Staphylococcus aureus (MRSA) infection has become the most common cause of skin and soft tissue infections seen in metropolitan emergency departments, and half-century-old therapeutic approaches are being revisited to combat it, according to two studies in the Aug. 17 New England Journal of Medicine.
More than three-fourths of all skin and soft tissue infections treated at 11 university-affiliated emergency departments in 2004 were colonized with S. aureus and 59% overall were methicillin resistant, reported Gregory J. Moran, M.D., of the Olive View-UCLA Medical Center here, and colleagues.
The areas with the highest MRSA prevalence were Atlanta at 72%; Kansas City, at 74%; and Charlotte, N.C., at 68%. Los Angeles had a 51% MRSA prevalence while New York was the lowest at 15%.
By comparison, MRSA was an uncommon cause of skin and soft tissue infections prior to 2000, hovering around 3% depending on geographic location.
Dr. Moran and colleagues in the EMERGEncy ID Net Study Group prospectively looked at these infections at the 11 metropolitan centers. Specimens from the 422 adult patients were cultured at each hospital using standard methods and those that were found to be S. aureus were sent to the CDC for further characterization.
In an editorial, M. Lindsay Grayson, M.D., of the University of Melbourne, Australia, called the investigation by Dr. Moran and colleagues a “landmark study,” detailed the “amazing extent to which community-associated MRSA, particularly the USA300 clone, has spread through the U.S. population.”
He commented that treatment options are “weighted in favor of surgical drainage as the priority intervention–a concept better known to clinicians before the days of penicillin.”
USA300 isolates accounted for nearly all of the MRSA isolates(97%) while a single strain called USA300-0114 accounted for 74% of these. Almost all of the MRSA samples tested had SSCmec type IV characteristic of community-associated MRSA and the Panton-Valentine leukocidin (pvl) toxin gene, which has been associated with spontaneous skin and soft-tissue infections and necrotizing pneumonia.
Although resistant to the antibiotic methicillin, most of the MRSA specimens were still susceptible to clindamycin (95%) and fluoroquinolones (60%). All were vulnerable to rifampin and trimethoprim-sulfamethoxazole. Only 6% were susceptible to erythromycin.
Most patients were treated with the combination of incision and drainage and antibiotics (66%). Another 10% received only antibiotics and 19% underwent incision and drainage alone. Five percent received neither. The most commonly used antibiotics were antistaphylococcal penicillin and cephalosporin (64%).
The antibiotic used conflicted with the results of the susceptibility testing for 57% of patients. However, consistent with other reports, this did not make any significant difference in the outcome.
“This absence of an association?suggests that most simple skin abscesses, even when caused by MRSA, can be cured with adequate drainage alone,” Dr. Moran and colleagues wrote.
Other basic practices recommended by Dr. Grayson included surgical drainage and debulking of abscesses, use of older narrow-spectrum antimicrobial agents, and prevention of transmission by:
The study was supported by a cooperative arrangement with the CDC. Some of the authors of Dr. Moran’s study reported funding from Schering-Plough, Pfizer, Aventis, Cubist, and Otho-McNeil. Dr. Grayson has had funding from Pfizer and Bayer.
An open-label study in the same NEJM issue found that the antibiotic Cubicin (daptomycin) was as good as standard therapy for bacteremia and endocarditis caused by S. aureus.
Cubicin itself is an older treatment that was abandoned because to toxicity issues more than a decade ago, but is back with a new dosing regimen, reported Vance G. Fowler, Jr., M.D., M.H.S., of Duke, and colleagues.
Of the 124 patients randomized to 6 mg/kg of body weight of Cubicin intravenously each day, 44.2% had a “successful” outcome after the endpoint of 42 days compared with 41.7% of the 122 patients who received low-dose gentamicin plus an antistaphylococcal penicillin or vancomycin.
Complicated bacteremia, right-sided endocarditis, and MRSA infections had similar success rates.
The Cubicin group had higher, though insignificantly so, rates of microbiologic failure (19 versus 11 patients, P= 0.17) and of adverse events that led to treatment failure due to the discontinuation of therapy (17 versus 8, P= 0.06) compared with the standard therapy group.
However, standard therapy led to more clinically significant renal dysfunction compared with Cubicin (26.3% versus 11.0%, P= 0.004).
Dr. Grayson noted several limitations of the study including the fact that “although this trial was randomized, it was an open-label study with the clinical outcome assessed in a blinded fashion. Thus, although bias in assessing treatment efficacy may have been controlled, bias in reporting and acting on adverse events was not.”
He also noted that “although the study aimed to recruit 90 patients who could be evaluated in each treatment group (to assess a potential treatment difference of ?20% ), only 79 patients who received Cubicin and 60 patients who received standard therapy were fully evaluated.”
He pointed out that 32% of 19 patients who received Cubicin and had microbiologic treatment failure had isolates that had developed resistance to daptomycin Thus, he said “clinicians should be aware that if treatment with this agent appears to be failing, the emergence of resistance should be carefully assessed.”
He concluded that despite these important concerns and caveats, Cubicin “may be cautiously considered as a potential treatment option for some patients with S. aureus bacteremia.”
The Cubicin study was supported by Cubist Pharmaceuticals and Dr Fowler and many of the co-authors reported having served as consultants for Cubist Pharmaceuticals. In addition, two of the co-authors are employees of Cubist Pharmaceuticals.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


