
Adverse Drug Reaction, Purple Glove Syndrome. The lawyers and attorneys at our firm are currently offering free case evaluations to victims of Purple Glove Syndrome. This serious condition, which causes the legs/and or arms to become painfully swollen and turn purple, can become serious, and lead to the amputation of the affected limb. If you […]
Adverse Drug Reaction, Purple Glove Syndrome. The lawyers and attorneys at our firm are currently offering free case evaluations to victims of Purple Glove Syndrome. This serious condition, which causes the legs/and or arms to become painfully swollen and turn purple, can become serious, and lead to the amputation of the affected limb. If you or a loved one suffered from this condition, we urge you to contact one of our ‘Purple Glove Syndrome’ lawyers right away to discuss your case.
In September 2008, the Food & Drug Administration (FDA) released a list of 20 drugs that were being investigated because of reported adverse reactions. One of the medications on the roster was the antiepileptic drug Dilantin (phenytoin). The FDA said there had been reports that patients receiving intravenous phenytoin had developed ‘Purple Glove Syndrome’.
The FDA did not say how many such reports it had received, but our Purple Glove Syndrome lawyers believe a great number of people may have been injured by this drug. A 1998 study found that of 152 patients receiving intravenous Dilantin (phenytoin), 9 (about 5.9%) developed Purple Glove Syndrome. That study, which was conducted by researchers at the Mayo Clinic, concluded that ‘Purple Glove Syndrome’ is not rare and that elderly patients and individuals receiving large, multiple doses of intravenous phenytoin are most likely to develop the disorder.
Phenytoin acts to dampen the unwanted, runaway brain activity seen in seizure by reducing electrical conductance among brain cells by stabilizing the inactive state of voltage gated sodium channels. Aside from seizures, it is an option in the treatment of trigeminal neuralgia as well as certain cardiac arrhythmias.
In the US, phenytoin is marketed as Phenytek and Dilantin (including Dilantin Kapseals, Dilantin-125 and Dilantin Infatabs). Intravenous phenytoin has been in use since 1956. This treatment is linked to several other serious side effects, including hypotension, arrhythmias and toxicity.
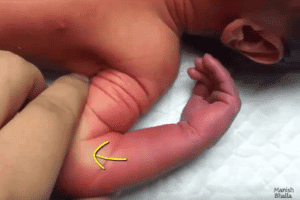
‘Purple Glove Syndrome’ was so named because of the discoloration of the skin that often accompanies the disorder. This discoloration is accompanied by pain and edema distal to the site of intravenous administration of phenytoin.
Generally, ‘Purple Glove Syndrome’ starts as a pale blue or dark purple discoloration which appears around the intravenous insertion site 2 to 12 hours after the administration of the drug. Progression occurs during the next 12 to 16 hours as developing edema and continued discoloration spread around all sides of the extremity.
It can take 2-4 weeks for a case of Purple Glove Syndrome to heal. If a patient is fortunate, the discoloration and edema will gradually fade. While the disorder can resolve without incident, our Purple Glove Syndrome lawyers are aware of several cases resulting in necrosis (death of tissue) that have been reported.
No one knows exactly what causes Purple Glove Syndrome. In some cases, it appears to be related to the reaction of the interstitial tissue to leakage of the highly alkaline pH of phenytoin injection. However, not all cases of Purple Glove Syndrome are preceded by such leakage. It is thought that this varying reaction is related to the formulation of phenytoin injection.
If a patient exhibits symptoms of Purple Glove Syndrome, there are a number of steps healthcare providers can take to lessen its severity. First and foremost, the administration of phenytoin must be stopped. The application of heat to the affected area can help to relieve pain and to redistribute the phenytoin. The affected extremity will also be elevated to lessen edema.
Severe cases of ‘Purple Glove Syndrome’ require more intervention. This could include emergency surgery to relieve pressure and restore blood flow. The most severe cases of Purple Glove Syndrome can lead to amputation of the affected limb. For this reason, therapeutic nursing interventions aimed at maximizing healing and promoting comfort are essential.
If you or a loved one suffered from Purple Glove Syndrome following intravenous administration of phenytoin, you have valuable legal rights. Please fill out our online form, or call 1-800 YOURLAWYER (1-800-968-7529) to discuss your case with one of our experienced Purple Glove Syndrome Lawyers as soon as possible.


