
According to a recall announcement from the U.S. Food and Drug Administration, HAMILTON-C6 Intensive Care Ventilators have been identified as a Class I Recall. Class I Recalls are the most severe type of recall because the product is linked to serious injuries and fatalities. The HAMILTON-C6 Intensive Care Ventilators were distributed from August 31, 2017, until May […]
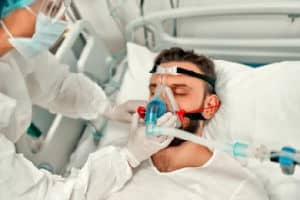
Hamilton-C6 Intensive Care Ventilator Lawsuits
According to a recall announcement from the U.S. Food and Drug Administration, HAMILTON-C6 Intensive Care Ventilators have been identified as a Class I Recall. Class I Recalls are the most severe type of recall because the product is linked to serious injuries and fatalities. The HAMILTON-C6 Intensive Care Ventilators were distributed from August 31, 2017, until May 20, 2022. The infant, intensive care ventilators are being recalled because the ventilator’s status indicator board can become loose, allowing water to enter and causing the unit to fail. Should the ventilator fail, the infant could suffer a lack of oxygen, leading to brain injury or death.
The HAMILTON-C6 Intensive Care Ventilator provides breathing support or mechanical ventilation to neonatal infants, babies, children, and adult patients. The ventilators provide ventilation invasively through a tube in the nose, mouth, or trachea and non-invasively through a breathing mask that is placed over the patient’s nose, mouth, or face. HAMILTON-C6 ventilators are used by trained healthcare personnel under the supervision and direction of a physician.
Hamilton Medical AG has initiated the HAMILTON-C6 Intensive Care Ventilator recall after receiving several customer complaints. The recall states that the device’s hardware issue involves the ventilator’s status indicator board. When the status indicator board becomes loose, it permits liquid to leak onto the ventilator’s main board. Then the ventilator can sustain a technical fault reverting to a safety ventilation mode or ambient state, causing the patient to breathe ambient room air without assistance or support from the ventilator. If an alternative source of ventilation is not provided immediately, the patient may suffer interrupted ventilation leading to brain injury (hypoxia), carbon dioxide buildup in the blood (hypercarbia), serious injuries, or death. So far, there have been more than 128 complaints associated with this issue.
Hamilton Medical AG published a Field Safety Notice to service engineers, healthcare facilities, and distributors concerning the recall. The Field Safety Notice recommends the following actions:
Medical professionals and patients affected by these devices are urged to notify the FDA’s MedWatch by online form, Fax, or regular mail.
Did you or a loved one sustain harm due to a recalled medical device? Parker Waichman LLP helps those who have suffered product injuries receive full monetary compensation. Trust your case with our product injury lawsuit lawyers. For a free consultation, contact our law firm today by using our live chat or calling 1-800-YOUR-LAWYER (1-800-968-7529).


