
DePuy ASR Defective Hip Judgment. Johnson & Johnson’s (J&J) DePuy facility defectively designed their metal-on-metal hip implants, according to a California jury, in the first of over 10,000 defective hip lawsuits to go to trial. The hip implant in question, the DePuy ASR hip, has been notorious for causing serious injury to individuals after only […]
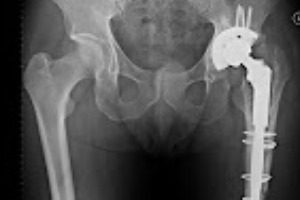
DePuy ASR Defective Hip Judgment. Johnson & Johnson’s (J&J) DePuy facility defectively designed their metal-on-metal hip implants, according to a California jury, in the first of over 10,000 defective hip lawsuits to go to trial.
The hip implant in question, the DePuy ASR hip, has been notorious for causing serious injury to individuals after only a few years of use. At the end of the trial, the jury awarded the plaintiff, Loren “Bill” Kransky, a total of $8.3 million dollars in damages for the injuries he sustained from the defective hip. Jurors said J&J was negligent in failing to warn healthcare providers of the risks associated with their defective hip implant.
In August of 2010, J&J recalled 93,000 of its hip implants, when data showed 12 percent failed within 5 years of being implanted. Other shocking figures came from Australian regulatory authorities that reported a 44 percent failure rate within 7 years. Due to the widespread damage caused by the defective hip implants, analysts are estimating the subsequent lawsuits could cost J&J billions of dollars to resolve.
Mr. Kransky had injuries very similar to the other thousands of injured victims who have either filed a lawsuit against J&J or have consulted legal counsel regarding their injuries. After having the device implanted, Mr. Kransky suffered from dislocations, pain, and had to incur multiple surgeries to correct the defective implant.
Kransky’s lawyers argued that DePuy failed to test the device adequately before selling it in the U.S., hid surgeon complaints from the FDA, and studied a redesign of the ASR hip before scrapping that effort in 2008. After the trial Kransky’s lawyer said “This is the first day of reckoning for DePuy.”
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


