
July 29, 2011: A panel of medical experts urges the U.S. Food & Drug Administration (FDA) to abandon the medical device approval system since it offers no assurance of safety, as reported by ABC News. hip Are you a victim of the DePuy ASR hip implant recall? Thousands of people across the U.S. were implanted […]

July 29, 2011: A panel of medical experts urges the U.S. Food & Drug Administration (FDA) to abandon the medical device approval system since it offers no assurance of safety, as reported by ABC News.
hip
Are you a victim of the DePuy ASR hip implant recall? Thousands of people across the U.S. were implanted with DePuy Orthopaedic’s defective ASR hip replacement system between 2005 and 2010, when it was finally recalled. Many of those patients experienced the premature failure of their DePuy ASR hip implant within just few years of their surgery. Even those patients who haven’t yet experienced problems with their DePuy ASR hip implant face an uncertain future.
Our DePuy ASR hip implant lawyers are offering a free lawsuit consultation to anyone injured by this defective device. If you underwent hip replacement surgery since 2005, and have suffered from unexplained hip pain, thigh pain or groin pain, pain with walking, pain rising from a seated position, or pain with weight bearing, you may be the victim of a failed DePuy ASR hip implant. We urge you to contact one of our DePuy ASR hip implant lawyers today to protect your legal rights.
The ways in which the DePuy ASR hip implant can fail include: Loosening of the various components in the implant;
Symptoms suffered by patients who have been implanted with the DePuy ASR hip implant were outlined to include:
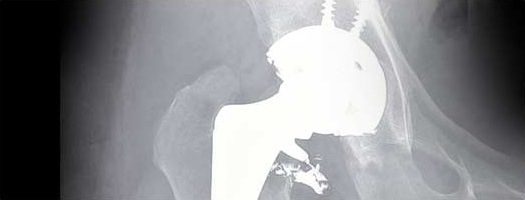
The DePuy ASR Acetabular Cup System first came on the market in 2005. It was cleared by the U.S. Food & Drug Administration (FDA) via a process known as 510(k) approval. This process allows a manufacturer to obtain market approval with very little clinical testing of the device – including no human testing – if a manufacturer can prove it is “substantially similar” to another product already on the market. In 2010, an internal FDA review – ironically released shortly after the DePuy ASR hip implant recall was issued – found numerous flaws with the 510(k) process, prompting the agency to consider changing it.
In 2009, DePuy had already announced it would be phasing out the ASR hip implant, ostensibly because of low sales. But by 2008, the FDA had received more than 300 reports of problems with the hip implant, most involving premature failure that required revision surgery. A hip implant should last for roughly 15 years, but many DePuy ASR hip implant recipients were undergoing new surgeries just a few years after implantation.
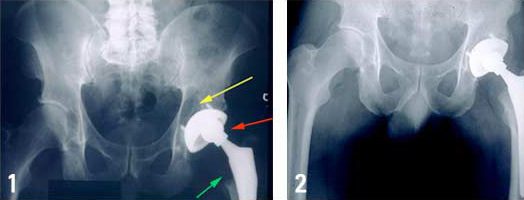
Then in a letter dated March 6, 2010, DePuy warned doctors that recently analyzed data from Australia suggested that the ASR had a higher-than-expected failure rate when used in traditional hip replacement on certain types of patient. According to the letter, patients of small stature, a group that typically includes women and patients with weak bones, faced the highest risk.
Finally, in August 2010, DePuy issued a worldwide recall for the ASR XL Acetabular Hip Replacement System after data from the National Joint Registry of England and Wales showed that 1 out of every 8 patients (12%-13%) who had received the devices had to undergo revision surgery within five years of receiving it. By then, more than 93,000 patients worldwide were fitted with an ASR hip implant. It is believed that roughly a third of those were patients in the U.S.
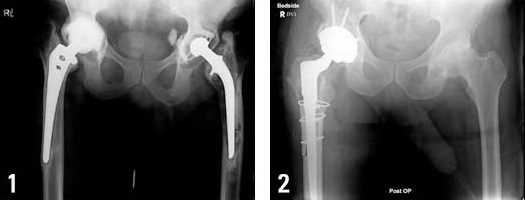 The DePuy ASR hip implant is a metal-on-metal device made of chromium and cobalt, and consists of a cup that’s implanted into the hip with a ball joint that connects to the leg. According to a New York Times report published in March 2010, metal-on-metal hip implants have been used in about one-third of the approximately 250,000 hip replacements performed annually in this country. However, many of the nation’s leading orthopedic surgeons have reduced or stopped use of these devices because of concerns that they can cause severe tissue and bone damage.
The DePuy ASR hip implant is a metal-on-metal device made of chromium and cobalt, and consists of a cup that’s implanted into the hip with a ball joint that connects to the leg. According to a New York Times report published in March 2010, metal-on-metal hip implants have been used in about one-third of the approximately 250,000 hip replacements performed annually in this country. However, many of the nation’s leading orthopedic surgeons have reduced or stopped use of these devices because of concerns that they can cause severe tissue and bone damage.
It is believed that many of the complications linked to the DePuy ASR XL Acetabular hip implant are caused by wearing of the metal components, which can allow metal shavings to make their way into patients’ bloodstreams, leading to tissue breakdown, bone loss, and even the formation of non-cancerous tumors. The shedding of metal shavings can cause cobalt poisoning, a disorder that, if left untreated, can put patients at risk of tinnitus (ringing in the ears), vertigo, deafness, blindness, optic nerve atrophy, convulsions, headaches, peripheral neuropathy, cardiomyopathy, and hypothyroidism. Even patients who are not experiencing issues with their DePuy ASR hip implant may still be at risk for future complications. DePuy ASR hip implant patients have been advised to have their blood cobalt level
tested, and such individuals may require medical monitoring for years to come.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


