
Questions About Device Safety of IVC Filters Continuos to Grow. Litigation involving inferior vena cava (IVC) filters continues to grow, as questions about device safety persist. Plaintiffs allege that the IVC filters, which are placed inside a major blood vessel to stop a blood clot from reaching the heart or lungs, resulted in serious injuries. […]
Questions About Device Safety of IVC Filters Continuos to Grow. Litigation involving inferior vena cava (IVC) filters continues to grow, as questions about device safety persist.
Plaintiffs allege that the IVC filters, which are placed inside a major blood vessel to stop a blood clot from reaching the heart or lungs, resulted in serious injuries.
Multidistrict litigations (MDLs) have been established for lawsuits against C.R. Bard and Cook Medical. MDLs are created when there are numerous lawsuits with similar allegations, as centralizing the cases helps make legal proceedings more efficient.
Doctors place IVC filters in patients at risk for pulmonary embolism, a potentially fatal condition where a blood clot reaches the lungs. The risk of pulmonary embolism is higher in patients with conditions such as venous thromboembolism or deep vein thrombosis.
Some IVC filters are retrievable, others permanent. A number of the lawsuits allege that injury occurred during a botched removal, where the filter breaks and perforates.
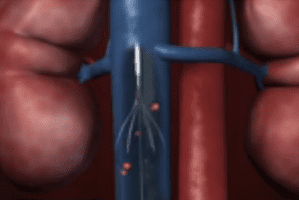
During both implantation and removal, the filter is placed via a catheter inserted into a large vein such as in the neck or groin. The filter attaches itself to the walls of the inferior vena cava, a major vein carrying deoxygenated blood from the lower and middle parts of the body to the right side of the heart.
An NBC investigation shed light on the safety of IVC filters manufactured by Bard, and questioned whether the device maker acted appropriately. According to the news report, the Recovery filter was linked to 27 deaths over the course of a decade.
Two studies focused on IVC filters were published recently in the journal Seminars in Interventional Radiology. In one study, researchers analyzed the rate of injuries based on the type of IVC filter used.
The goal was to determine which patients would benefit from retrieval versus just follow-up. The study found that filters categorized as purely conical were associated with the highest risk of penetration (90-100%).
The risk of IVC thrombosis was highest with cylindrical or umbrella elements (30-50%) and Conical Bard filters were associated with the highest reported risks of fracture (40%).
In another study, researchers focused on medical device litigation involving IVC filters, citing a rising number of lawsuits in recent years.
The authors state that both Cook’s and Bard’s MDLs share common questions of facts, specifically whether the filter design and manufacturing practices made them unreasonably prone to complications.
“The resolution of these cases will add to a larger legal debate concerning how much legal protection the 1976 Medical Device Amendments should offer firms from tort liability.
As a specialty that often relies on medical devices, it is not only important for interventional radiologists to have a general understanding of medical device litigation but also to reflect upon the approaches to informed consent regarding these devices.” the authors state.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


