
Flextome Angioplasty Device Recalled. The U.S. Food and Drug Administration (FDA) and Boston Scientific Corporation have notified healthcare professionals regarding the class 1 recall of an angioplasty device (Flextome Cutting Balloon Device and Monorail Delivery System), due to the potential for distal catheter shaft separation during device withdrawal. The device consists of a surgical balloon with […]
Flextome Angioplasty Device Recalled. The U.S. Food and Drug Administration (FDA) and Boston Scientific Corporation have notified healthcare professionals regarding the class 1 recall of an angioplasty device (Flextome Cutting Balloon Device and Monorail Delivery System), due to the potential for distal catheter shaft separation during device withdrawal.
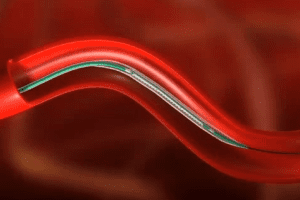
The device consists of a surgical balloon with microsurgical blades (microtomes) attached to the sides. It is inserted via catheter into the artery, and then expanded to allow the blades to cut through fatty deposits and widen the blocked coronary artery.
Shaft separation may prolong the procedure or necessitate more in-depth surgery to remove the remaining fragment from the artery, according to an alert sent Friday from MedWatch, the FDA’s safety information and adverse event reporting program.
According to a company news release, 8 cases of shaft separation have been reported, 3 of which required surgery.
The FDA notes that the recall does not affect patients who have already received treatment, because the problem occurs during the angioplasty procedure.
Healthcare professionals are requested to cease using the product, and to contact the company at 1-800-811-3211 to arrange for returns. The recall affects approximately 40,000 devices worldwide.
Adverse events related to use of the angioplasty device should be reported to the FDA’s MedWatch reporting program by phone at 1-800-FDA-1088, by fax at 1-800-FDA-0178, online at http://www.fda.gov/medwatch, or by mail to 5600 Fishers Lane, Rockville, MD 20852-9787.
The personal injury attorneys at Parker Waichman offer free, no-obligation case evaluations. For more information, fill out our online form or call 1-800-YOURLAWYER (1-800-968-7529).


