
Clear Cell Adenocarcinoma Injury Lawsuits. Clear cell adenocarcinoma is a rare form of cervical and vaginal cancer and was the first health problem determined to be associated with DES (diethylstilbestrol) exposure. DES (diethylstilbestrol) is a synthetic form of estrogen that was prescribed between 1940 and 1971 to help women who encountered complications during pregnancy. Women […]
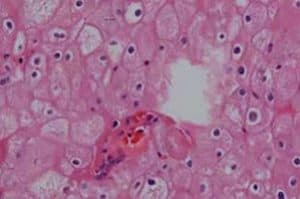
Clear Cell Adenocarcinoma Injury Lawsuits. Clear cell adenocarcinoma is a rare form of cervical and vaginal cancer and was the first health problem determined to be associated with DES (diethylstilbestrol) exposure. DES (diethylstilbestrol) is a synthetic form of estrogen that was prescribed between 1940 and 1971 to help women who encountered complications during pregnancy. Women who used DES may have a slightly increased risk of breast cancer.
Before 1971, clear cell adenocarcinoma of the vagina or cervix was a rare disease, diagnosed primarily in women over the age of seventy. In 1971, doctors documented several cases of this cancer in young women whose mothers were prescribed DES during pregnancy. This led the FDA to ban the use of DES during pregnancy in 1971.
Children and grandchildren of women who took DES during pregnancy are at an increased risk of developing significant injuries ranging from rare cancers to genital abnormalities. All DES daughters (women whose mothers took DES while pregnant with them) have a risk of about 1 in 1,000 of being diagnosed with clear cell adenocarcinoma.
DES daughters have a higher risk for ectopic pregnancy, miscarriage, and preterm labor and delivery. Most DES daughters can become pregnant and carry their babies to term. However, because of the above risks, all DES daughters (whether they have had previous normal pregnancies or not) require high-risk obstetric care and early confirmation of pregnancy. DES daughters should have their pregnancies confirmed by a health care provider as soon as pregnancy is suspected, and they should be seen more frequently throughout their pregnancies.
If you have been diagnosed with clear cell adenocarcinoma or if your mother or grandmother took DES while pregnant, fill out the form at the right for a free case evaluation with a drug side effects lawyer or call us at 1-800-YOURLAWYER (1-800-968-7529).


