
Health Canada has announced new warnings for 5-alpha reductase inhibitors, or 5-ARIs, regarding there association with an aggressive form of prostate cancer. The new warnings will apply to Proscar (finasteride 5mg), Propecia (finasteride 1mg), Avodart (dutasteride) and Jalyn (dutasteride and tamsulosin). Proscar, Avodart and Jalyn are all used to treat enlarged prostate, while Propecia is […]
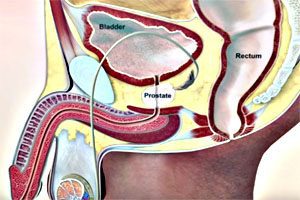
Health Canada has announced new warnings for 5-alpha reductase inhibitors, or 5-ARIs, regarding there association with an aggressive form of prostate cancer.
The new warnings will apply to Proscar (finasteride 5mg), Propecia (finasteride 1mg), Avodart (dutasteride) and Jalyn (dutasteride and tamsulosin). Proscar, Avodart and Jalyn are all used to treat enlarged prostate, while Propecia is used to treat male pattern hair loss.
According to Health Canada, the new safety information is based on Health Canada’s review of two large international clinical trials, the Prostate Cancer Prevention Trial (PCPT) and the Reduction by Dutasteride of Prostate Cancer Events (REDUCE) trial.
The trials showed that the long-term daily use (over 4 years) of finasteride (5 mg) and dutasteride in men aged 50 years and older was associated with a small but statistically significant increased risk of high-grade prostate cancer.
The 1 mg finasteride strength (Propecia) was not included in these trials but a potential risk has not been ruled out, according to Health Canada.
High-grade prostate cancer is an aggressive type of prostate cancer that grows and spreads more quickly than low-grade prostate cancer.
Last summer, the U.S. Food & Drug Administration (FDA) issued a Drug Safety Communication notifying the public that the “Warnings and Precautions” sections of the labels of all FDA-approved 5-ARIs was being modified to include new information that they may increase the risk of high-grade prostate cancer.
The FDA communication was prompted by its review of the same studies cited in the Health Canada announcement.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations.
For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


