
On April 5, 2017, the United States Judicial Panel on Multidistrict Litigation transferred six actions in three districts pending against medical device maker Stryker into a multidistrict litigation (MDL) in the District of Massachusetts. The plaintiffs’ claims focus on the performance Stryker’s LFIT V40 cobalt-chromium device, in particular the alleged propensity of the device to […]
 On April 5, 2017, the United States Judicial Panel on Multidistrict Litigation transferred six actions in three districts pending against medical device maker Stryker into a multidistrict litigation (MDL) in the District of Massachusetts.
On April 5, 2017, the United States Judicial Panel on Multidistrict Litigation transferred six actions in three districts pending against medical device maker Stryker into a multidistrict litigation (MDL) in the District of Massachusetts.
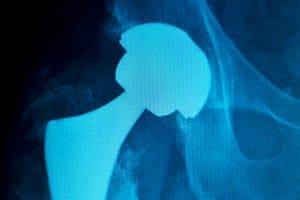
The plaintiffs’ claims focus on the performance Stryker’s LFIT V40 cobalt-chromium device, in particular the alleged propensity of the device to cause corrosion at the junction when the heads are paired with femoral stems made from different alloys (such as HOC’s proprietary TM F, which is an alloy of titanium, molybdenum, zirconium and iron). This corrosion allegedly leads to failure of the implant or to other serious health consequences that necessitate surgery to remove and replace the implants.
The Stryker LFIT is not the only hip device that is the subject of litigation. Parker Waichman notes that thousands of lawsuits have been filed against the makers of metal-on-metal hip replacement hips including DePuy Orthopaedics (a subsidiary of Johnson & Johnson), Biomet, Wright Medical Technology, and Smith & Nephew.
In August 2016, Stryker Howmedica Osteonics Corp. recalled 42,519 defective hips. The company sent a letter to orthopedic surgeons advising them of a “higher than expected” incidence of taper lock failure for certain sizes of its LFIT Anatomic CoCr V40 Femoral Heads.
At about the same time, Health Canada, the Canadian public health agency, issued a recall of the Stryker LFIT V40 and the Australian Government Department of Health published a safety alert about the devices.
The LFIT femoral head has been marketed for use with a variety of femoral stems. Some medical literature says the problem arises when stems made of titanium or TMZF titanium alloy are combined with cobalt-chromium alloy femoral head and taper devices.
The pairing of the different metal alloys results in fretting and corrosion at the junction where the femoral head connects to the femoral stem. Fretting refers to wear and corrosion damage to the contact surfaces. The damage is caused by repeated motion, as, for example, in walking and normal movement of a hip-implant recipient. Fretting damages the metal’s surface layer, producing increased surface roughness and micropits, which reduce the strength of the components.
Corrosion at this junction has led to the release of metal particles into the bloodstream and the tissue and bone around the joint. Patients have suffered toxic metallosis, tissue death, bone death, and elevated levels of cobalt and chromium in the blood. Stryker hip recipients have experienced complications including difficulty walking, inflammation, adverse local tissue reaction, joint instability, hip dislocation, broken bones, and the need for revision surgery.
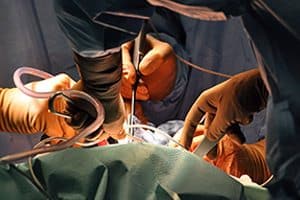 When a hip replacement patient undergoes revision surgery—surgery to remove and replace a defective implant—the patient faces all the risks he or she faced in the original surgery. The patient may experience post-surgical complications or develop an infection. Recovery from hip replacement involves extensive physical therapy to regain strength and range of motion. Repeat surgeries are often not as successful as the original hip replacement, especially when the defective hip device has further damaged the joint.
When a hip replacement patient undergoes revision surgery—surgery to remove and replace a defective implant—the patient faces all the risks he or she faced in the original surgery. The patient may experience post-surgical complications or develop an infection. Recovery from hip replacement involves extensive physical therapy to regain strength and range of motion. Repeat surgeries are often not as successful as the original hip replacement, especially when the defective hip device has further damaged the joint.
The medical community had high expectations for metal-on-metal hips when they came to market. Surgeons thought the metal components would prove durable than older designs devices made of plastic, ceramic or a combination of materials. Earlier hip devices generally had a useful life of about 15 years, which meant that many hip recipients would need to undergo replacement again. With more people having hip replacement at younger ages, surgeons and patients were looking for hip device that would withstand the rigors of physically demanding professions and allow the recipient to participate in sports and leisure activities without pain. Manufacturers promoted metal-on-metal hips as the long-waited advance in hip devices.
Critics say the problems with the Stryker LFIT and other metal-on-metal hips could have been revealed through adequate testing. But many metal-on-metal hips came to market through a streamlined device approval process that allowed the manufacturer to avoid testing. The U.S. Food and Drug Administration’s 510(k) device clearance pathway allows a manufacturer to bring a device to market without clinical trials if the device is considered substantially similar to a device already on the market. But many experts note that even small difference in materials or design can make a big difference in a device’s safety or effectiveness. Once a device is implanted in the body, it can be difficult to correct problems or repair damage the device causes. All devices should be subject to testing before they are allowed on the market.
If you or someone you know has needed revision surgery or suffered other side effects from a Smith & Nephew hip device or other metal-on-metal hip, Parker Waichman LLP can provide a free, no-obligation case evaluation. To consult one of our experienced attorneys, fill out the online form or call 1-800-YOURLAWYER (1-800-968-7529).


