
The Food and Drug Administration (FDA) has issued a warning to tattoo artists and consumers to avoid certain tattoo inks. On August 4, A Thousand Virgins Corp. in Miami (City of Industry), Florida recalled all lots of tattoo grey wash inks, G1, G2 and G3, labeled Lot #129, expiration date 1/16, because of microbiological contamination. FDA […]
FDA Issues Warning about Contaminated Tattoo Inks
The Food and Drug Administration (FDA) has issued a warning to tattoo artists and consumers to avoid certain tattoo inks.
On August 4, A Thousand Virgins Corp. in Miami (City of Industry), Florida recalled all lots of tattoo grey wash inks, G1, G2 and G3, labeled Lot #129, expiration date 1/16, because of microbiological contamination.
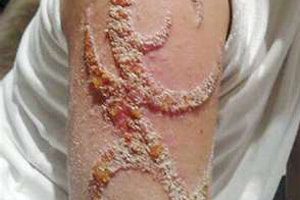
FDA laboratory testing found microbial contamination in the inks. The inks are contaminated with Mycobacterium chelonae organisms and the molds Cryptococcus albidus and members of the Penicillium genus. Use of these products may cause bacterial infection and can lead to sepsis, a potentially life-threatening complication of an infection.
The inks were manufactured, sold and distributed by A Thousand Virgins Corp. The G1, G2 and G3 grey wash tattoo inks were distributed nationwide in individual bottles and in kits and may be available for sale from international and online distributors. Despite the recall, the FDA is concerned that artists and consumers are continuing to use these contaminated inks from their current stock.
The FDA is warning tattoo ink manufacturers, tattoo artists and consumers not to use the tattoo inks that have been recalled. Artists and consumers who purchase tattoo inks or individuals who seek tattooing should check the ink bottles to see if they are included in the recall. Anyone who finds inks subject to recall should place the closed bottles of ink into a plastic bag, sealing or tying off the bag to prevent leakage. Place the first bag into a second bag and tie off this bag separately. Before disposing of the inks, check with local waste management authorities for requirements for proper disposal.
Consumers with questions about the inks may contact the company at 1-866-829-4659 from Monday to Friday between the hours of 9am to 5pm EST.
In July 2014, White and Blue Lion, Inc., of City of Industry, California, issued a similar ink recall. According to Linda Katz, M.D., M.P.H., director of FDA’s Office of Cosmetics and Colors, these inks could cause infection. “FDA has confirmed one case of skin infection involving a consumer that used this company’s tattoo products.”
“Tattooing poses a risk of infection to anyone,” Katz warns, “but the risk is particularly high for those with pre-existing heart or circulatory disease, diabetes or compromised immune systems.” Katz notes that injecting contaminated ink into the skin or using contaminated needles may result in infections at the site of the tattoo. Signs of localized infection include redness, swelling, weeping wounds, blemishes, or excessive pain at the site. Anyone who experiences these signs should seek medical care right away. Even after a localized infection has healed, the area may be permanently scarred. An infection that is left untreated or inadequately treated could spread through the bloodstream (a process known as sepsis).
The FDA encourages consumers and tattoo artists to take precautions to avoid infections. The agency encourages consumers and tattoo artists to report adverse events through the MedWatch Safety Information and Adverse Event Reporting Program so that the FDA can investigate and prevent others from becoming infected.


