
The U.S. Food and Drug Administration (FDA) has put its highest-risk label on Medtronic’s recall of its Duet external drainage and monitoring systems. The product is used to externally drain and monitor cerebrospinal fluid and monitors intracranial pressure; it is part of Medtronic’s Neurosurgery division. The products include: Interlink Injection Sites, Catalog Number 46913 SmartSite […]
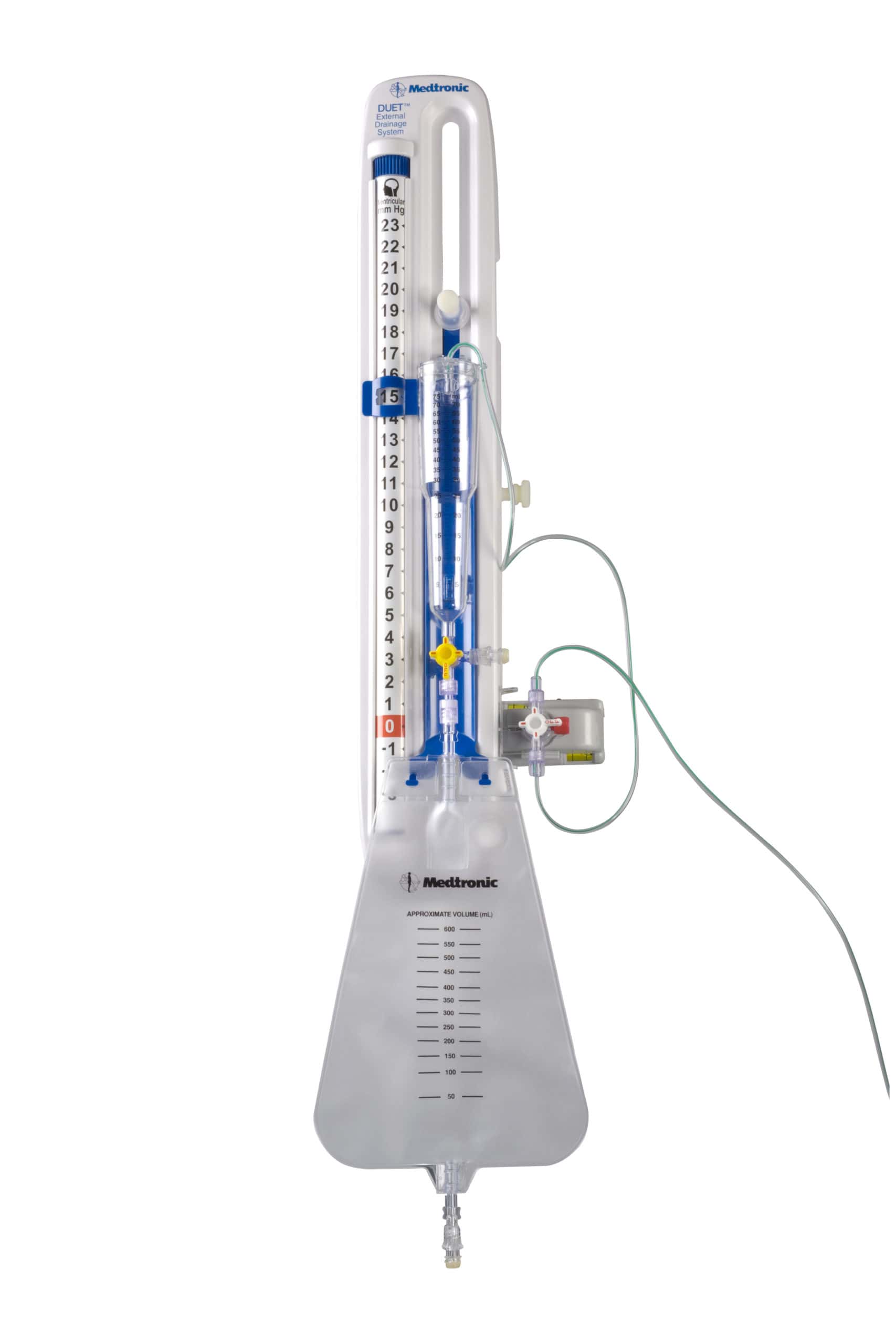 The U.S. Food and Drug Administration (FDA) has put its highest-risk label on Medtronic’s recall of its Duet external drainage and monitoring systems. The product is used to externally drain and monitor cerebrospinal fluid and monitors intracranial pressure; it is part of Medtronic’s Neurosurgery division. The products include:
The U.S. Food and Drug Administration (FDA) has put its highest-risk label on Medtronic’s recall of its Duet external drainage and monitoring systems. The product is used to externally drain and monitor cerebrospinal fluid and monitors intracranial pressure; it is part of Medtronic’s Neurosurgery division. The products include:
Interlink Injection Sites, Catalog Number 46913
According to the recall notice, the devices were recalled because the patient line tubing can be separated from the patient line connectors; this is more likely occur during frequent handling and include connections were injections or sampling occur often. The notice stated that “The device failure may result in air within the skull (pneumocephalus), infection (such as meningitis, ventriculitis, encephalitis) and over/under drainage of the CSF that may contribute to serious adverse health consequences, including death.”
The recalled systems were manufactured between March 15, 2013 and February 28, 2014. They were distributed between April 10, 2013 and May 19, 2014. An Urgent Device Recall letter was sent out to customers last month.
A Medtronic spokesperson told Mass Device that the company received 35 reports of adverse events between September 2013 and April 2014. No deaths have been reported thus far.
A Class I label means that exposure to the recalled device could lead to serious injury or death.


