
Mentor Corp., a unit of Johnson & Johnson that makes transvaginal mesh devices, has reached a settlement just weeks before a trial was scheduled to begin in a transvaginal mesh device case. The company was accused of making defective pelvic mesh implants that cause painful infections and other complication, Law360 reports. The trial was scheduled to begin […]
J&J Reach Settlement in Bellwether Pelvic Mesh Case
Mentor Corp., a unit of Johnson & Johnson that makes transvaginal mesh devices, has reached a settlement just weeks before a trial was scheduled to begin in a transvaginal mesh device case.
The company was accused of making defective pelvic mesh implants that cause painful infections and other complication, Law360 reports. The trial was scheduled to begin on November 30.
Last week, Mentor filed settlement documents in Georgia federal court settling a case with a couple who brought a complaint over the company’s ObTape sling. The case was among six cases designated as bellwethers by U.S. District Judge Clay D. Land, Law360 reports.
The bellwether trial is an approach sometimes used when large numbers of plaintiffs have similar claims, creating a large caseload. A group of plaintiffs is chosen to represent all the plaintiffs. Representative cases go to trial and the results can be an indication of likely results in other trials. In some instances, bellwether cases provide the basis for reaching a settlement in the remaining cases.
In the ObTape cases, Mentor faces claims of negligence, fraudulent concealment, breaches of express and implied warranties and failure to warn, Law360 reports. In one of the potential bellwether cases, the judge initially threw out the plaintiff’s fraudulent concealment claim but vacated that order in September.
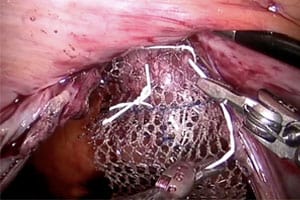
Pelvic mesh—also called transvaginal mesh—is used to treat pelvic organ prolapse (POP) and stress urinary incontinence (SUI) in women. According to the Food and Drug Administration (FDA), POP occurs when the internal structures that support the bladder, uterus and bowel, become weak, stretched, or torn and the organs drop from their normal position and bulge (prolapse) into the vagina. Women with POP experience symptoms including pain and disruption of sexual, bladder, and bowel functions. Incontinence can be embarrassing and have a tremendous negative impact on a woman’s life. But many women who have transvaginal mesh devices implanted to correct POP and SUI experience complications from the devices. The complications include erosion of the mesh through the vaginal lining, infection, pain, urinary problems, and recurrence of prolapse or incontinence. The FDA has also received reports of bowel, bladder, and blood vessel perforation during insertion of the mesh device. Many of the women need additional surgeries to correct the problems.
The settled case was brought by a woman who was implanted with the ObTape device in 2004, and her husband. By 2011, she said she had suffered infections, abscesses and scarring that required multiple surgeries, according to Law360.
In 2014, the FDA issued two proposed orders to address the health risks associated with transvaginal mesh. These orders would reclassify surgical mesh devices for transvaginal POP from a moderate-risk device (Class 2) to a high-risk device (Class 3) and require manufacturers to submit a premarket approval (PMA) application for the agency to evaluate safety and effectiveness.
In 2014, the day before the U.S. Judicial Panel on Multidistrict Litigation consolidated the ObTape suits into a multidistrict litigation (MDL), the company settled 111 claims. Mentor had settled another proposed bellwether case in 2013, shortly before trial. There are multidistrict litigations in New Jersey and West Virginia involving mesh manufacturers C.R. Bard, Boston Scientific, Johnson & Johnson’s Ethicon division, Coloplast, and Cook Medical.


