
The national law firm, Parker Waichman LLP, just filed another lawsuit over the Mirena® intrauterine device (IUD). The lawsuit alleges that the Mirena® IUD caused a Pennsylvania woman substantial pain and suffering, perforating her uterus and migrating to the area of the sacrum, located at the base of the spine. The Mirena® IUD was then […]
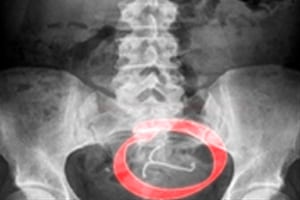 The national law firm, Parker Waichman LLP, just filed another lawsuit over the Mirena® intrauterine device (IUD). The lawsuit alleges that the Mirena® IUD caused a Pennsylvania woman substantial pain and suffering, perforating her uterus and migrating to the area of the sacrum, located at the base of the spine. The Mirena® IUD was then found behind the woman’s left ovary when she underwent laparoscopic surgery to have the device removed.
The national law firm, Parker Waichman LLP, just filed another lawsuit over the Mirena® intrauterine device (IUD). The lawsuit alleges that the Mirena® IUD caused a Pennsylvania woman substantial pain and suffering, perforating her uterus and migrating to the area of the sacrum, located at the base of the spine. The Mirena® IUD was then found behind the woman’s left ovary when she underwent laparoscopic surgery to have the device removed.
The lawsuit was filed on November 30th in the Superior Court of New Jersey Law Division, Morris County. Bayer Healthcare Pharmaceuticals has been named as the defendant. (Docket No. MRS-L-2927-12)
According to the complaint, the woman was implanted with the Mirena® IUD in July 2009. She appeared to tolerate the initial procedure well, and neither she nor her doctor had reason to believe that the device perforated her uterus. During a follow-up visit in 2010, her doctor was unable to locate the IUD’s strings and was also unable to locate the device by ultrasound or hysteroscopy. An x-ray revealed that the Mirena® IUD had migrated to the area of the sacrum, which is the large triangular bone at the base of the spine. The patient had to undergo laparoscopic surgery in January 2011 to retrieve the Mirena® IUD, which was found behind her left ovary. During the procedure, omentum adhesions and an ovarian cyst were also found.
The lawsuit alleges that Bayer Healthcare Pharmaceuticals failed to adequately warn about the risks of using Mirena® IUD. According to the complaint, Bayer only warns about the risks of uterine perforation when the device is first implanted; safety information does not warn about spontaneous migration risks, which is what allegedly occurred in this patient. The suit alleges that the Bayer’s negligent and wrongful actions caused this woman to suffer severe and permanent physical injuries, substantial pain and suffering, and economic loss due to medical expenses and lost wages.
In 2009, the Department of Health and Human Services’ Division of Drug Marketing, Advertising, and Communications (DDMAC) warned Bayer about misleading statements made concerning the marketing of the Mirena® IUD. The DDMAC found that claims made during the company’s “Simple Style” program overstated the benefits of Mirena® while failing to mention its risks. The program’s script claimed that Mirena® would help women increase libido as well as help them “look and feel great.” Presenters also claimed that Mirena® did not require any type of compliance to a monthly routine. The DDMAC deemed all of these claims as false and reprimanded Bayer for failing to mention side effects such as weight gain, acne and breast pain or tenderness.
As we’ve explained, the Mirena® IUD was approved by the U.S. Food and Drug Administration (FDA) in 2000 as an intrauterine contraceptive. The T-shaped Mirena® device releases a synthetic progestogen known as levonorgestrel into the uterus to prevent pregnancy, although Bayer admits that the exact mechanism remains unknown. In 2009, the Mirena® IUD was also approved to treat heavy menstrual bleeding in women who use IUDs. Mirena® is recommended for women who have had at least one child, and is indicated for up to five years of use. The device is used in over two million women in the United States and more than 15 million women worldwide, according to the FDA.
The agency has said that Mirena® has been associated with serious complications such as ectopic pregnancy, intrauterine pregnancy, group A streptococcal sepsis, pelvic inflammatory disease (PID), device embedment in the uterine wall, and uterine wall and cervix perforation. Side effects also include bleeding and spotting at irregular intervals, headaches, ovarian cysts, vaginitis, pain during menstruation, pelvic pain, and breast tenderness.
As we’ve previously mentioned, Canadian health regulators reminded healthcare providers there of Mirena®’s risks of uterine perforation.


