
DePuy Orthopaedics and its parent, Johnson & Johnson, continue to be hit with lawsuits over recalled metal-on-metal DePuy ASR hip implants. Just yesterday, the national law firm of Parker Waichman LLP announced it had filed yet another DePuy ASR hip implant lawsuit, this time on behalf of a Virginia resident who claims one of the […]
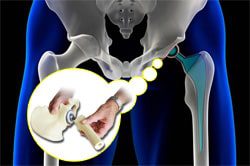 DePuy Orthopaedics and its parent, Johnson & Johnson, continue to be hit with lawsuits over recalled metal-on-metal DePuy ASR hip implants. Just yesterday, the national law firm of Parker Waichman LLP announced it had filed yet another DePuy ASR hip implant lawsuit, this time on behalf of a Virginia resident who claims one of the defective implants caused her to suffer from elevated chromium and cobalt levels.
DePuy Orthopaedics and its parent, Johnson & Johnson, continue to be hit with lawsuits over recalled metal-on-metal DePuy ASR hip implants. Just yesterday, the national law firm of Parker Waichman LLP announced it had filed yet another DePuy ASR hip implant lawsuit, this time on behalf of a Virginia resident who claims one of the defective implants caused her to suffer from elevated chromium and cobalt levels.
The lawsuit was filed on April 12, in the DePuy ASR hip implant multidistrict litigation currently underway in the U.S. District Court, Northern District of Ohio. The complaint was among several Parker Waichman LLP filed in the litigation that day.
According to the lawsuit, the plaintiff, a 62-year-old woman from Ferrum, Virginia, received a DePuy ASR Hip Implant on October 10, 2007. By February 2011, the Plaintiff was suffering from pain, as well as elevated chromium and cobalt levels due to her metal-on-metal DePuy ASR Hip Implant. The plaintiff is asking the court for medical monitoring, as well as compensatory and punitive damages.
DePuy ASR Hip Implant Recall
DePuy issued a worldwide recall of the ASR Hip Resurfacing System and ASR Acetabular System in August 2010, after data from the National Joint Registry of England and Wales showed that 1 out of every 8 patients (12%-13%) who had received the devices had to undergo revision surgery within five years of receiving it. By the time the implants were pulled off the market, more than 93,000 people worldwide had received one of the devices, according to a statement from Parker Waichman LLP.
Metal-on-Metal Hip Implant Dangers
The ASR hip implants are metal-on-metal devices, a class currently being reviewed by the U.S. Food & Drug Administration (FDA). Last May, the agency directed 21 companies that market all-metal hip replacements, including DePuy, to conduct post-market studies of their products to determine if they were shedding dangerous amounts of metallic debris in patients. The FDA recently announced that its Orthopaedic and Rehabilitation Devices Panel will meet over June 27 and 28 to discuss the risks and benefits of metal-on-metal hip systems, as well as make potential patient and practitioner recommendations for their use.
It is believed the metal debris shed by metal-on-metal hip implants can lead to serious side effects, including:
In March, a large study published in The Lancet showed that there is a 6.2 percent chance patients with all-metal hips will need a replacement within five years. “Metal-on-metal stemmed articulations give poor implant survival compared with other options and should not be implanted,” the study authors wrote. The month prior, a report in the British Medical Journal revealed that metal-on-metal hip implant manufacturers were aware of mounting evidence linking the devices to serious, long-term health consequences, but for years failed to warn the public about these dangers. The report warned that hundreds of thousands of people around the world may have been exposed to dangerously high levels of toxic and potentially cancer-causing metals from failing metal-on-metal hip implants.
Since the DePuy ASR hip implant recall, DePuy and other manufacturers have been named in lawsuits over their metal-on-metal hip replacement devices. In addtion to the DePuy ASR hip implant litigation in Ohio. Claims involving a metal-on-metal version of DePuy’s Pinnacle hip implant have been consolidated in a multidistrict litigation in Texas. In February, a multidistrict litigation was established in the U.S. District Court for the Northern District of Georgia for lawsuits involving the all-metal Wright Conserve Hip Replacement System. Several lawsuits are also pending in the U.S. over Biomet metal-on-metal hip implants.


