
Medtronic Inc.’s published studies for its Infuse bone graft product may have downplayed serious side effects, according to an analysis published last year in The Spine Journal. The study, which drew on U.S. Food & Drug Administration (FDA) documents and other data sources, suggests that up to half of patients receiving Infuse may experience adverse […]
 Medtronic Inc.’s published studies for its Infuse bone graft product may have downplayed serious side effects, according to an analysis published last year in The Spine Journal. The study, which drew on U.S. Food & Drug Administration (FDA) documents and other data sources, suggests that up to half of patients receiving Infuse may experience adverse events, including:
Medtronic Inc.’s published studies for its Infuse bone graft product may have downplayed serious side effects, according to an analysis published last year in The Spine Journal. The study, which drew on U.S. Food & Drug Administration (FDA) documents and other data sources, suggests that up to half of patients receiving Infuse may experience adverse events, including:
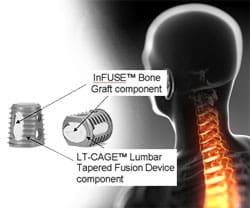
Medtronic Infuse is a synthetic form of recombinant human Bone Morphogenetic Protein (rhBMP-2) that is approved for use in a certain type of spinal surgery. The U.S. Food & Drug Administration (FDA) has only approved Infuse graft for use in single-level anterior lumbar fusion. However, it is known that the majority of procedures that utilize Infuse are off-label. While doctors can use an FDA-approved product in anyway they see fit, manufacturers are legally barred from promoting those uses. Medtronic Infuse lawsuits allege that the company illegally promoted unapproved uses of Infuse. In 2008, the FDA warned that Infuse and similar products had caused serious and potentially life-threatening problems when they were used off-label in cervical spine (neck) surgeries.
The Spine Journal study, conducted by Eugene Carragee, MD, of Stanford University’s outpatient clinic in Redwood City, Calif., and colleagues, investigated 13 reports of studies funded by product manufacturer Medtronic and published from 2000 to 2009. According to a report from MedPage Today, the studies purported to have found no adverse events attributed to Infuse.
“In the larger trials, there is evidence in each trial that rhBMP2 complications may be common and may be serious; but in each publication these were unreported,” Carragee and his colleagues wrote.
Carragee’s analysis also raised concerns about the financial ties between Medtronic and the researchers who conducted the 13 studies, according to MedPage Today. Some apparently had associations with the device maker valued at more than $10 million.
In an accompanying editorial, Carragee and his colleagues slammed the disclosure policies of the journals that published the Infuse studies. For example, they were critical of one article in Spine, which stated: “‘The manuscript submitted does not contain information about medical device(s)/drug(s). Institutional funds were received in support of this work. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.”
But according to Carragee and colleagues, “Even the most cursory review shows that this was all about devices and drugs used in an off label manner and reported by authors who, by conservative estimates, have tens of millions of dollars of financial association with the sponsor.”
After The Spine Journal published Carragee’s study, Medtronic took the unusual step of commissioning researchers at Yale University to review Infuse clinical trials. Results from that review are expected later this year.


