
In a June 2015 article in Respiratory Medicine Case Reports, researchers from Southern Illinois University and the University of Texas Medical Branch at Galveston concluded their case report with the recommendation that further research be conducted to determine the long-term safety and effectiveness of IVC (inferior vena cava) filters, inserted to prevent blood clots from […]
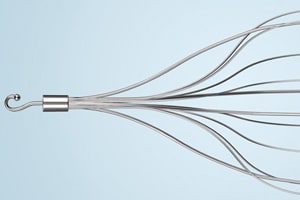 In a June 2015 article in Respiratory Medicine Case Reports, researchers from Southern Illinois University and the University of Texas Medical Branch at Galveston concluded their case report with the recommendation that further research be conducted to determine the long-term safety and effectiveness of IVC (inferior vena cava) filters, inserted to prevent blood clots from traveling to the heart and lungs.
In a June 2015 article in Respiratory Medicine Case Reports, researchers from Southern Illinois University and the University of Texas Medical Branch at Galveston concluded their case report with the recommendation that further research be conducted to determine the long-term safety and effectiveness of IVC (inferior vena cava) filters, inserted to prevent blood clots from traveling to the heart and lungs.
The IVC filter is inserted into the inferior vena cava—the body’s main vein—to help keep clots from being carried into the lungs. This procedure is typically reserved for people who cannot take anticoagulant drugs or when anticoagulant drugs don’t work well enough or fast enough, according to the Mayo Clinic.
People at risk for venous blood clots include those who have already had venous blood clots or pulmonary embolism. Other risk factors include heart disease, smoking, obesity, certain cancers, chemotherapy, prolonged immobility (for example after surgery, heart attack, or leg fracture), or sitting in a cramped position for a long period. Pregnancy also increases the risk, likely because of the weight of the baby pressing on veins in the pelvis, slowing blood return from the legs, according to the Mayo Clinic.
But IVC filters can lead to complications, including filter migration, filter occlusion (blockage), filter misplacement, and filter fracture with fragment embolization.
Medical device maker C.R. Bard, a leading maker of IVC filters, replaced a defective blood-clot filter line (Recovery) associated with more than two dozen deaths and hundreds of injuries with a version (G2) that is now linked to at least 12 deaths and hundreds of problems. NBC News reports that Bard knew the G2 filters had similar flaws soon after the line was put on the market.
NBC News obtained records indicating that within four months of receiving Food and Drug Administration (FDA) clearance to sell the G2 filter, Bard was expressing concern about reports of failures for G2 filters. Bard did not recall the G2 filters, but kept them on the market until 2010, and sold more than 160,000 G2 filters in that time.
Each year, about 250,000 people have IVC filters implanted to prevent blood clots from moving into the heart and lungs, where they could be fatal. Some filters are intended to remain in the body permanently, while others are removed when the clot danger has passed. When pieces of a filter break off and migrates, the patient can experience severe pain, bleeding, further embolus, and other life-threatening complications.
Bard faces numerous lawsuits over injuries associated with its IVC filter lines. In August 2015, the lawsuits were consolidated into a multidistrict litigation in the District of Arizona.
Though the doctors did not attempt to remove the broken filter strut from the patient discussed in Respiratory Medicine Case Reports, and she remained asymptomatic, the authors suggested the need for “strict compliance with the current guidelines of IVC filter insertion, adequate follow up, and retrieval of the filters when they are no longer indicated.”


