
The Oregon Department of Justice just obtained a $4 million settlement over a lawsuit brought over Johnson & Johnson’s DePuy Orthopaedics Inc. metal-on-metal hip implant device. The case involved allegations concerning the way in which DePuy marketed the medical device. The Oregon Department of Justice alleged that the device maker sold a line of metal-on-metal hip […]
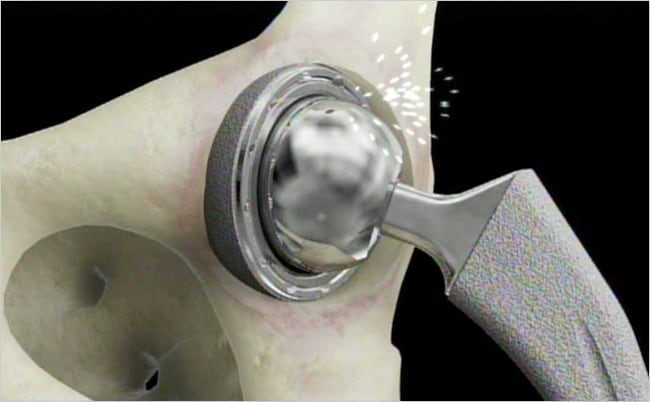 The Oregon Department of Justice just obtained a $4 million settlement over a lawsuit brought over Johnson & Johnson’s DePuy Orthopaedics Inc. metal-on-metal hip implant device. The case involved allegations concerning the way in which DePuy marketed the medical device.
The Oregon Department of Justice just obtained a $4 million settlement over a lawsuit brought over Johnson & Johnson’s DePuy Orthopaedics Inc. metal-on-metal hip implant device. The case involved allegations concerning the way in which DePuy marketed the medical device.
The Oregon Department of Justice alleged that the device maker sold a line of metal-on-metal hip implant devices that were prone to early failure, according to Oregon Live. In Oregon, 432 devices were sold and Oregon is the first government lawsuit that was brought over the devices.
To date, DePuy has settled thousands of lawsuits brought over its ASR XL hips, which were globally recalled in 2010. All-metal hips have been tied to high and premature failure rates and a number of alleged, adverse medical reactions, such as increased blood metal ion levels and metal poisoning; dislocations; pain; fracture; difficulty ambulating, rising, standing, and balancing; noise emanating from the joint; and pseudotumors, to name just some. Patients have also alleged debris from the chromium and cobalt hip device have led to tissue death and increased blood metal ion levels, according to a prior Bloomberg.com report.
According to Oregon Live, DePuy took advantage of a controversial U.S. Food and Drug Administration’s (FDA) process. The metal-on-metal hip devices were approved under the FDA’s fast-tracked 510(k) approval route. Under this process, device makers are able to bypass clinical testing of their products if a device is found to be substantially similar to an already-approved device. While the DePuy devices were released to market under the 510(k), studies reveal that metal-on-metal devices fail far more frequently than similar, but non-all-metal devices.
This particular Oregon-led action only involved the state, noted Oregon Live, which noted that, in other cases, Oregon has led a number of multi-states actions. The Unfair Trade Practice Act settlement was filed in Multnomah County Circuit Court. Although DePuy admits no wrongdoing, the settlement does includes an agreement by DePuy that it would not engage in deceptive marketing in the future, say VendorsBay.
“Oregonians in need of a hip replacement deserve to know that the artificial hip they are contemplating in fact has the qualities, and benefits, that a company advertises,” said Oregon Attorney General Ellen Rosenblum, according to Oregon Live. “Doctors also need to know that the products they suggest to their patients meet certain standards; and no company should be permitted to exploit that basic tenet.” This settlement amounts to approximately $10,000 per device sold in Oregon.
A DePuy spokesperson issued a statement that indicated, in part, “As the agreement states, this is a settlement of a disputed matter. There is no admission of any legal violation, and DePuy denies any such violation.”
In a corporate filing it made in May, DePuy’s parent company, Johnson & Johnson, disclosed the investigation in Oregon and also disclosed another, similar multi-state investigation into DePuy. The U.S. Department of Justice is also investigating, wrote Oregon Live.


