
Risk Of Injury With Soft Tissue Filler. The Food and Drug Administration (FDA) has issued a safety communication warning of the risk of injury if soft tissue fillers—also known as dermal fillers, wrinkle fillers, or injectable facial implants—are accidentally injected into a blood vessel. The FDA issued the alert to warn health care providers and […]
Risk Of Injury With Soft Tissue Filler. The Food and Drug Administration (FDA) has issued a safety communication warning of the risk of injury if soft tissue fillers—also known as dermal fillers, wrinkle fillers, or injectable facial implants—are accidentally injected into a blood vessel.
The FDA issued the alert to warn health care providers and consumers about the possibility of rare, but serious, injuries that may occur if a soft tissue filler is unintentionally injected into blood vessels in the face.
The soft filler can block blood vessels and restrict blood supply to tissues. Sometimes this can result in embolization: the filler material travels to other parts of the body and can cause vision impairment, blindness, stroke and damage and/or death of the skin (necrosis) and underlying facial structures.
Soft tissue fillers “can create a smoother or fuller appearance of the face,” according to the FDA. They are FDA-approved to reduce the appearance of wrinkles or to augment lips or cheeks. Dermatologists, plastic surgeons, cosmetic surgeons, dentists, and other medical providers treat patients using soft tissue fillers.
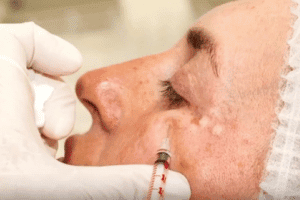
The soft tissue filler is injected directly into a treatment area. The FDA explains that results depend on the patient’s overall health and skin condition, the skill of the health care provider, the location of the injection, and the type of filler used. Patients may need more than one injection to get the desired smoothing/filling effect. Fillers should be injected only by health care providers who have appropriate training and experience and who are knowledgeable about the anatomy at and around the injection site.
The FDA has reviewed information that suggests unintentional injection of soft tissue fillers into blood vessels in the face can result in rare, but serious side effects. Unintentional injections into blood vessels may occur at injection sites anywhere on the face, but in its review of literature and adverse event reports, the agency found that blood vessel blockage is more likely to occur in facial injection sites: the skin between the eyebrows and nose (glabella), in and around the nose, forehead, and around the eyes (periorbital region).
The FDA stresses that before deciding to have soft tissue filler injections, the individual should speak the health care provider about appropriate injection sites and the risks associated with the procedure. Approved uses for particular fillers vary depending on the product and the patient should make sure the appropriate product will be used.
The health care provider should immediately stop the injection if a patient exhibits any signs or symptoms associated with injection into a blood vessel, such as changes in vision, signs of a stroke, white appearance (or blanching) of the skin, or unusual pain during or shortly after the procedure.
The patient should seek immediate medical attention if he or she develops symptoms such as unusual pain, vision changes, a white appearance of skin near the injection site, or any signs of a stroke. These include sudden difficulty speaking, numbness or weakness in the face, arms, or legs, difficulty walking, face drooping, severe headache, dizziness, or confusion during or shortly after the procedure.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


