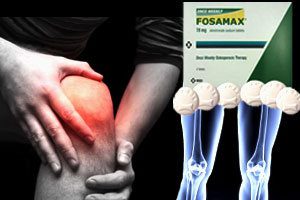
Reclast Lawsuit | Side Effects : Femur Fractures, Osteonecrosis of the Jaw (Dead Jaw Syndrome), Esophageal Cancer
Reclast Side Effects. Did you suffer a serious side effect from Reclast ( Zoledronic Acid )? Reclast is a bisphosphonate sold by Novartis Pharmaceuticals Corporation. The drug is administered intravenously [...]