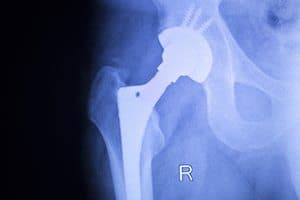
MoM Hip Replacement Recipients to Monitor and Follow-Up
The Medicines and Healthcare Products Regulatory Agency is calling for all metal-on-metal (MoM) hip replacement recipients to schedule follow-up appointments to monitor and detect adverse soft tissue reactions. The recommendation, [...]