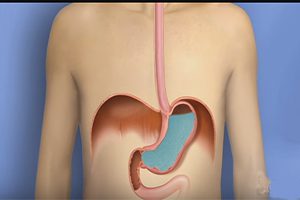
Takeda Faces New Lawsuit Alleging Prevacid Caused Kidney Injuries
Prevacid Lawsuit Alleges Heartburn Medication Caused Kidney Injury and Kidney Disease. Takeda Pharmaceuticals is facing a new lawsuit alleging that its popular heartburn drug Prevacid caused kidney injuries. The [...]