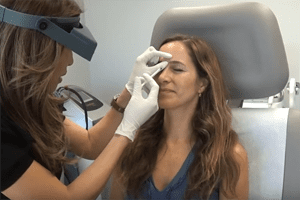
Botox injections It Could Be Dangerous Domestic health officials are warning hundreds of doctors in the U.S. about the dangers of importing Botox injections from Canada, saying they could be counterfeit and dangerous for their patients.
According to a Fox News report from an affiliate in Atlanta, the Food and Drug Administration recently warned at least 350 separate medical practices in the U.S. about the dangers of Botox being imported from Canada, specifically one major supplier that does much of its business online under myriad titles.
Canada Drugs has been shipping mass quantities of Botox injections to the U.S., presumably where they’re being administered to patients here. Many of these suspect vials are being used for cosmetic procedures to temporarily reduce or eliminate skin wrinkles. The FDA says there is no guarantee that these Botox injections are real, safe, or effective and that people receiving them in a procedure to reverse their signs of aging may be putting themselves at risk of grave illness.
The wholesale pharmacy Canada Drugs operates several sites that offer discounts on common prescription drugs and they’re readily available to anyone willing to take such a risk but in the U.S., the FDA considers these drugs illegal as they have not been passed through the agency’s approval process. Therefore, they say, the imported vials of Botox can not be guaranteed to be safe and could cause serious injuries or death if they’re dispensed to patients.
The FDA sent letters to 350 medical practices
The FDA sent letters to 350 medical practices late last year warning them about their purchases of Botox injections from a Canada Drugs outlet. The agency went so far as to post the names of those practices on its Web site so the public knows which doctors were possibly dispensing unregulated and unauthorized vials of Botox at their locations.
This warning from the FDA was the fifth of that kind in the last year, all concerning the illegal import of drugs from foreign suppliers, presumably done by doctors to cut some of their costs. Fox reports that any practice advertising lower-than-normal prices for Botox services may be those hawking their illegally-imported drugs. Prospective Botox recipients should not hesitate to ask questions of their physicians before receiving a treatment, specifically where the doctor purchased their injections.
Need Legal Help Regarding Botox imported?
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).
[sc name=”post-footers”]
Our Botox side effects attorneys are here to help you when you need it the most.