
DePuy Faces Lawsuits resulting from Faulty Adhesion of Knee Replacement Technology. In September of 2017, DePuy, a subsidiary of Johnson &Johnson, faced its first lawsuit resulting from the Attune Knee System. The lawsuit follows reports of adhesion problems experienced by patients who have been implanted with the Attune Knee System. The main reason the Attune […]
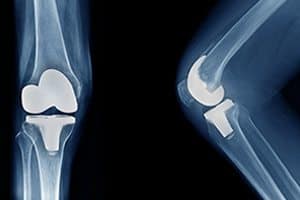
DePuy Faces Lawsuits resulting from Faulty Adhesion of Knee Replacement Technology. In September of 2017, DePuy, a subsidiary of Johnson &Johnson, faced its first lawsuit resulting from the Attune Knee System. The lawsuit follows reports of adhesion problems experienced by patients who have been implanted with the Attune Knee System. The main reason the Attune implants seem to be a failure to bond with the patient’s lower leg, which is technically referred to as “tibial baseplate failure.”
Knee replacement surgery is a procedure commonly performed on patients whose severe osteoarthritis causes them to have trouble with daily activities such as walking or climbing stairs. Over 600,000 knee replacements are performed in the United States each year.
A study printed in the Journal of Knee Surgery in the publication’s September 2017 issue indicated that the Attune system’s early failure rate was “unusually high.” The Food and Drug Administration (FDA) had received 232 reports about problems with Attune knee replacements. Hospital databases exhibited other reports of failures with the system. The study also indicated a likelihood of underreporting of complications as the result of radiographs not being able to assess loosening issues adequately.
According to nine orthopedic surgeons who contributed to the study, the substance used to bond the implant to the tibial component of the system was often failing to hold the implant in place. Once the tibial component becomes loose, the whole system becomes destabilized.
Patients whose Attune Knee System implants are failing might experience swelling, pain, decreased the range of motion, infection, and instability and loosening. Patients might notice instability in their knee when they stand or attempt to bear weight on their knee.
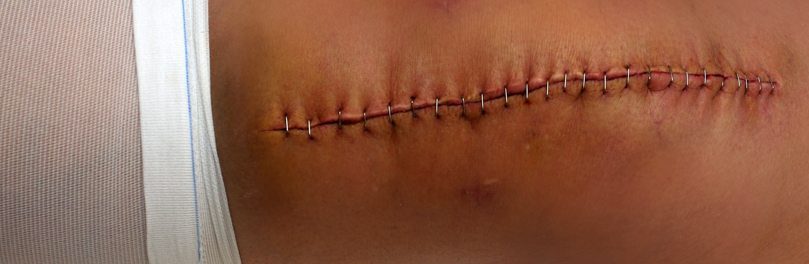
When patients’ knee replacement systems fail, the only option for them is to undergo surgery to remove the faulty system and replace it with a new one. Unfortunately, revision surgeries are more complicated than the initial procedure. This is in part because of many patient’s experience bone loss as a result of the device. This second surgery can be more painful dangerous, complex, and expensive than the first surgery, and the patient might also require a great deal of rehabilitation following the procedure.
Surgeons who have performed revision surgeries on recipients of Attune Knee replacements had reported that the implant not to have adhered to the bone at all, and in some instances, the removed implant looked the same as it had when it was initially removed from its packaging.
Any medical device will likely have some amount of failure. It is as of yet unclear how many DePuy knee replacement systems have been implanted in patients. Other forms of knee replacements, such as those made from ceramics, sometimes fail too, although at a rate of roughly 1 out of every 10,000. This number is considered low. While the number of failures of Attune systems is unsettling, it is difficult at this time to assess the relative risk of this device as compared to other similar products.
If you have experienced injury as the result of the DePuy Attune Knee Replacement System, you could be eligible to receive compensation. Call Parker Waichman LLP today at 1-800-YOURLAWYER (1-800-968-7529) for a free consultation with one of our experienced personal injury attorneys.


