
The FDA Announced that Painkillers Containing Benzocaine Are Unsafe For Children The U.S. Food and Drug Administration (FDA) recently announced that drug companies who manufacture over-the-counter (OTC) medications containing benzocaine must alter the way in which the companies market their products. The FDA announcement sternly warns drug manufacturers that marketing benzocaine as a method of […]
The U.S. Food and Drug Administration (FDA) recently announced that drug companies who manufacture over-the-counter (OTC) medications containing benzocaine must alter the way in which the companies market their products. The FDA announcement sternly warns drug manufacturers that marketing benzocaine as a method of relieving pain associated with teething endangered children two years of age and younger and insisted that companies stop advertising benzocaine for that purpose due to the persistent threat of methemoglobinemia, or reduced oxygen in the blood. The FDA threatened to take regulatory action if companies refused to comply with their recommendation. Additionally, the FDA insisted on new warning labels for benzocaine to inform consumers about the possible risks associated with using benzocaine. Furthermore, the FDA included prescription-based benzocaine medications which required additional box warnings on its labels.
Parker Waichman LLP is committed to holding drug companies accountable for their actions when someone suffers an injury or dies. Our clients expect the highest level of representation for their injuries, and that is precisely what they receive. Our clients feel that their losses are the direct result of drug companies refusing to look out for the consumer. When we take a case that involves products liability like a lawsuit over damages from taking benzocaine, our products liability lawyers fight hard for our clients to be compensated to the fullest extent possible.
The FDA issued its first warnings about the possible hazards of using benzocaine products to soothe teething pain approximately 12 years ago. The FDA followed their 2006 pronouncement up with additional warnings in 2011 and 2012. FDA regulators voiced their concern about the possibility of benzocaine causing methemoglobinemia in children. Kids two years old or younger proved to be the most vulnerable.
Incidents of methemoglobinemia in children were on the rise in 2012. The FDA stated that it received 29 complaints of methemoglobinemia occurring after the patient used a product containing benzocaine.
Nineteen of those complaints were from children. Most notably, 15 of the 19 cases involving children were children whose parents gave them products like Orajel, Baby Orajel, Anebsol, Chloraseptic, and other OTC temporary oral pain relievers.
Oral pain relievers have broader usage than gum pain. Companies market their benzocaine-based OTC drugs for relief of sore throats, toothaches, canker sores, cold sores, and other irritations of the mouth. The products come in sprays, liquids, solutions, lozenges, and gels.
Despite repeated warnings, the drug companies who make benzocaine-based products continued to advertise them to relieve gum pain associated with teething in small children. The trend continued into 2018. On May 21, 2018, the FDA sent a letter to the Chief Executive Officer of each drug maker that manufacturers any products containing benzocaine. In that correspondence, the FDA spelled out precisely why benzocaine is a threat to the public health. The FDA noted that between February 26, 2009, and October 6, 2017, it received 119 reports of methemoglobinemia associated with the use of benzocaine.
Four people out of the 119 died. One of the four was a four-month-old baby whose parents gave their child an OTC topical treatment for teething. The baby developed methemoglobinemia a short time after receiving the medicine. The FDA described only 3 out of the 119 cases as mild. In contrast, the FDA determined the remaining 116 cases to be “serious.”
The FDA proposed a multi-step plan to prevent additional deaths and illness due to benzocaine-induced methemoglobinemia. In the first step, the FDA advised that drug companies discontinue labeling its products to treat the symptoms associated with teething in children aged two or younger.
The FDA reasons that the amount of benzocaine a child ingests from an oral solution is disproportionate to what a child’s body can safely tolerate. Compounding the problem is those very young children are at a higher risk to develop methemoglobinemia than older children and adults. As a result, the FDA finally determined, after years of issuing warnings, that the risks associated with young children taking medications containing benzocaine outweighs any benefit the child could experience, especially when safer, holistic alternatives are readily available.
The second step the FDA advised drug makers to include additional warnings about benzocaine on its packaging. The FDA determined that the warning labels on benzocaine-based products must include additional language that the drug is not safe for use in children two years-of-age or younger and must not be used to soothe teething.
The third step in preventing benzocaine-induced methemoglobinemia according to the FDA is adding a warning that adults and children aged two or older are at risk of developing methemoglobinemia. The FDA recommends that such a notice be readily visible on the packaging. In its May 21, 2018 letter, the FDA demanded the addition of this warning. The agency asked the companies making OTC benzocaine medications to include this warning on their packaging four years earlier, but all declined to adopt that change. Now, the FDA is practically ordering it after 35 cases of benzocaine-induced methemoglobinemia came to the attention of the FDA.
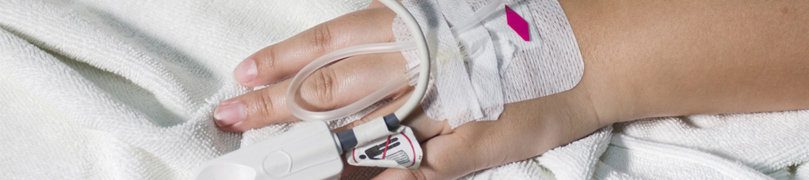
Methemoglobinemia can strike a person of any age. The research shows, however, that the youngest children are most vulnerable. Methemoglobinemia can be acquired, as in the case of benzocaine-induced Methemoglobinemia, or congenital. Essentially, Methemoglobinemia is a disease stemming from a lack of oxygen in the blood cells. The lack of freshly oxygenated blood cells deprives the body’s organs of the air they need to function correctly.
Methemoglobinemia is sometimes known as “baby-blue” syndrome. The symptoms of Methemoglobinemia include skin, fingers, nail beds, and lips turning blue. The formal medical terms for bluish skin is “cyanosis.”
Other symptoms include:
The treatment required for Methemoglobinemia depends on the severity of the case. Doctors will administer a drug called methylene blue to restore oxygen to the blood. If this fails to treat the condition, doctors must give the patient a blood transfusion.
Methemoglobinemia has potential long-term ramifications and can lead to respiratory diseases such as asthma, bronchitis, emphysema, and heart disease. Fortunately, most people recover from this severe medical complication.
Call Parker Waichman LLP today to ensure that you do not lose out on your chance to obtain legal justice for you. We have recovered over $2 billion in financial compensation for our clients. Also, we are fortunate to have received several awards bestowed upon us by our peers for relentless advocacy on behalf of our clients. Let us fight for you against the makers of benzocaine-based oral pain relievers if you or your child developed Methemoglobinemia from using benzocaine.
Consultations with Parker Waichman LLP’s Heartmate lawsuit lawyers are free-of-charge. Call 1-800-YOURLAWYER (1-800-968-7529) to schedule yours today. We are here 24/7 for you. Our benzocaine lawsuit lawyers have a tremendous amount of first-hand experience protecting our clients’ rights and maximizing their financial compensation.


