
Dangerous Health Problems May Be Linked to Limbrel. The United States Food and Drug Administration (FDA) has issued a warning for those taking the drug Limbrel. Around the beginning of September, the FDA started looking into reports of liver injuries and hypersensitivity pneumonitis, which is inflammation in the alveoli of the lungs, in patients […]
Dangerous Health Problems May Be Linked to Limbrel. The United States Food and Drug Administration (FDA) has issued a warning for those taking the drug Limbrel.
Around the beginning of September, the FDA started looking into reports of liver injuries and hypersensitivity pneumonitis, which is inflammation in the alveoli of the lungs, in patients who took Limbrel.
On November 30, 2017, the FDA expressed its concerns about Limbrel to Primus Pharmaceuticals. Although the FDA advised Primus Pharmaceuticals to voluntarily recall Limbrel at that time, the company failed to do so.
By early December, approximately 200 adverse events had been reported to the FDA. In around 30 of these reports, there was enough evidence for the FDA to conclude whether Limbrel was linked to the health complications that were being reported. Additional reports have been sent to the FDA.
The FDA sent a formal request to Primus Pharmaceuticals on December 18, 2017 that advised the company to recall Limbrel. The FDA believes that those who take Limbrel may suffer serious health complications, such as liver injury.
Limbrel comes in several doses. The FDA has recalled Limbrel 250 mg capsules (flavocoxid) as well as Limbrel250 capsules (250 mg flavocoxid with 50mg citrated zinc bisglycinate). Limbrel 500 mg capsules (flavocoxid) and Limbrel500 (500mg of flavocoxid with 50 mg citrated zinc bisglycinate) have also been recalled.
The FDA has asked medical professionals and consumers to use its MedWatch Adverse Event Reporting program to report adverse reactions or other issues with Limbrel.
Limbrel, according to Primus Pharmaceuticals, was advertised to help patients “manage the metabolic processes associated with osteoarthritis.” It comes in several different dosages. Some dosages contain only flavocoxid, whereas others also include zinc.
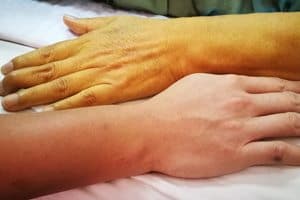
The FDA has reported that Limbrel may be associated with drug-induced liver injuries, as well as hypersensitivity pneumonitis.
According to the FDA, anyone who is currently taking Limbrel should stop taking the drug immediately.
Symptoms of liver issues may include fatigue, gastrointestinal problems, jaundice, and nausea.
With hypersensitivity pneumonitis, patients should watch out for weight loss, breathing issues, chronic bronchitis, chills, headache, fever, cough, and fatigue.
Anyone who has taken Limbrel and experiences these symptoms should report them to a medical professional.
Of course, if other adverse reactions are noted while taking Limbrel, these should be reported as well.
If you have experienced negative side effects from taking Limbrel, you should make note of these symptoms and report them to your medical professional as soon as possible.
Your medical professional may decide to continue monitoring your health for any other signs of harm caused by Limbrel. For example, you may undergo blood tests to determine if you have suffered any liver damage.
If it is determined that you were injured by Limbrel, you may be entitled to damages.
Every year, thousands of individuals suffer injuries caused by adverse reactions from drugs. Some of these injuries are life-threatening. In severe drug injury cases, individuals die due to complications caused by the substance.
To make a claim for monetary damages in a drug injury case, a claimant must support the elements of such a case with evidence. Drug injury claims are similar to other products liability claims, in that an individual alleges that using a product has resulted in an injury.
The elements of a drug injury case include the following:
If the claimant cannot prove one of these elements, the drug injury claim may fail.
Showing that a drug caused an injury can be difficult. After all, how are symptoms caused by the drug isolated from symptoms that may be caused by other conditions? For example, how do you prove if headaches are related to a defective drug, or if they are caused by sinus problems?
To prevail in a drug injury case, a claimant must show that he or she was taking the drug as instructed and that the drug is directly related to the injuries the claimant endured.
With the assistance of an experienced drug injury attorney, it is possible to provide strong evidence of drug injury claims. The following are types of evidence that are often used in drug injury cases:
So long as it is relevant to the case, many types of evidence may be used in drug injury claims.
Every drug injury case is different. Some individuals suffer relatively minor injuries that they recover from in a few days or weeks, whereas others may have permanent damage due to the defective drugs. The purpose of awarding damages in a drug injury case is to make the claimant “whole” again after suffering an injury. Types of damages that may be awarded in a drug injury case include:
An experienced drug injury attorney will make sure that as much evidence as possible is submitted to the court in support of the injured claimant’s damages.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


