
increasing number of patients implanted with the HeartMate II Last year, doctors at the Cleveland Clinic noticed that an increasing number of patients implanted with the HeartMate II pump device were developing potentially fatal blood clots soon after the device was implanted. This type of heart implant is used on patients in advanced stages of […]
 increasing number of patients implanted with the HeartMate II Last year, doctors at the Cleveland Clinic noticed that an increasing number of patients implanted with the HeartMate II pump device were developing potentially fatal blood clots soon after the device was implanted. This type of heart implant is used on patients in advanced stages of heart failure, such as former Vice President Dick Cheney, according to a New York Times report.
increasing number of patients implanted with the HeartMate II Last year, doctors at the Cleveland Clinic noticed that an increasing number of patients implanted with the HeartMate II pump device were developing potentially fatal blood clots soon after the device was implanted. This type of heart implant is used on patients in advanced stages of heart failure, such as former Vice President Dick Cheney, according to a New York Times report.
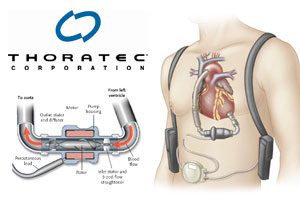
Manufactured by Thoratec Corp., the HeartMate II is a left ventricular assist device; it features a pump that pushes the patient’s blood through the heart on an ongoing basis.
This year, researchers from several hospitals, including the Cleveland Clinic, looked at hospital records and found that the incidence of blood clots among patients implanted with the HeartMate II after March 2011 was nearly four times greater than among patients implanted with the same device in previous years. In addition to the increase in blood clots, the clots also are occurring much more frequently than in the past, according to the New England study. After March 2011, the average duration prior to formation of a clot was 2.7 months, compared with 18.6 months in previous years.
Patients who developed pump-related clots either died or required emergency surgery for heart transplants or replacement devices.
In March 2013, the Cleveland Clinic informed both Thoratec and the U.S. Food and Drug Administration (FDA) about issues of blood clots over the implant, according to one researcher.
The New England Journal of Medicine posted a study about the device on its website in late November 2013 by researchers from the Cleveland Clinic, Duke University and Washington University in St. Louis. The study further notes that a preliminary analysis of data provided by a fourth hospital, the University of Pennsylvania, showed the same pattern of blood clot formation but was submitted too late for inclusion in the study.
The New England Journal of Medicine study found that the rate of pump-related blood clots, two months following the implant procedure, had risen to 8.4 percent after March 2011, from 2.2 percent in earlier years. Researchers suggested that the federally funded registry called Intermacs—which, as Thoratec noted in response, showed a smaller increase of 5% since March 2011, compared with 2 percent for earlier years—might not include all cases of pump-related blood clots, such as when a patient has an emergency heart transplant following the formation of a critical clot.
The FDA has issued a statement in which it notes that it is reviewing the findings of the New England study. It further notes that it “shares the authors’ concerns about the possibility of increased pump thrombosis.”
The sudden rise in pump-related blood clots noted in the study prompts questions regarding whether Thoratec Corp. had somehow modified the implant, either intentionally or accidentally, researchers noted. One said the leap in blood clots was probably device-related, “something as subtle as a change in software that affects pump flow or heat dissipation near a bearing.” Experts also add, according to the New York Times, that heart implants are prone to producing blood clots—which can form on a device’s surface—and note that the rate of blood clot formation can be affected by a variety of factors, including changes in use of blood-thinning drugs as well as the patient’s health.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


