
Unresolved Talks About Cook IVC Filter Litigation. The federal multidistrict litigation (MDL) involving Cook inferior vena cava (IVC) filters continues to move forward, with the first bellwether cases scheduled for 2017. The MDL was established in October 2014 in the Southern District of Indiana before U.S. District Court Judge Richard L. Young. More than 500 lawsuits […]
Unresolved Talks About Cook IVC Filter Litigation. The federal multidistrict litigation (MDL) involving Cook inferior vena cava (IVC) filters continues to move forward, with the first bellwether cases scheduled for 2017.
The MDL was established in October 2014 in the Southern District of Indiana before U.S. District Court Judge Richard L. Young. More than 500 lawsuits have been consolidated into the MDL, with plaintiffs alleging that the Cook Celect and Gunther Tulip IVC filters caused serious injuries. IVC filters are designed to catch blood clots before they reach the heart and lungs.
According to a court order filed June 29, 2016, attorneys for the plaintiffs and the defendants met with U.S. Magistrate Judge Tim Baker on June 17, 2016. The parties met in order discuss potential settlement terms.
However, they were unable to agree. Similar meetings may be held in the future in attempt to resolve the litigation without going to trial, but if these are also unsuccessful then the bellwether cases will take place.
Bellwether trials are the first lawsuits to go to trial. Their outcome is used to gauge how the remaining litigation will proceed, and may predict settlement terms.
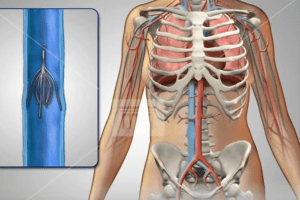
IVC filters are placed in the inferior vena cava, which transports blood from the lower body back to the heart and lungs. The small, cage-like device is meant to catch any blood clots from reaching the lungs, which would result in a life-threatening condition known as pulmonary embolism. However, there have been reports where IVC filters break, fracture and migrate to other parts of the body, leading to serious complications.
Nearly 1,000 adverse event reports related to IVC filters were submitted to the FDA between 2005 and 2010. In more than 300 cases, the IVC filter migrated to the heart or pulmonary artery, resulting in fluid buildup, arrhythmia or death. The filter detached and blocked an artery in roughly 150 cases and in 70 cases the device perforated artery walls or organs.
The FDA issued a safety alert in 2014 recommending that doctors remove IVC filters between 29 and 54 days after implantation as long as the patient is no longer at risk for pulmonary embolism. “The FDA has received reports of adverse events and product problems associated with IVC filters,” the agency said.
“Types of reports include device migration, filter fracture, embolization (movement of the entire filter or fracture fragments to the heart or lungs), perforation of the IVC and difficulty removing the device.”
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


