
Mini-Med Insulin Infusion Sets Worldwide Recall. In a September 11, 2017 press release, Medtronic announced a worldwide recall of some Mini-Med infusion sets used with Medtronic insulin pumps. The recall is related to a discontinued component in the infusion sets and does not include either the insulin pumps or glucose sensors used with the […]
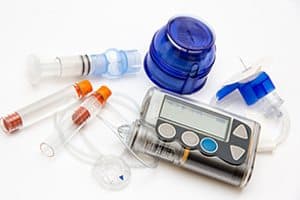
Mini-Med Insulin Infusion Sets Worldwide Recall. In a September 11, 2017 press release, Medtronic announced a worldwide recall of some Mini-Med infusion sets used with Medtronic insulin pumps.
The recall is related to a discontinued component in the infusion sets and does not include either the insulin pumps or glucose sensors used with the infusion sets.
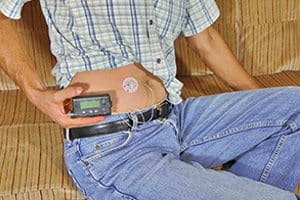
An insulin pump is an alternative to multiple daily injections of insulin by insulin syringes or insulin pens. The pump can deliver small amounts of insulin continuously for normal functions of the body or can deliver additional insulin on demand to match the food the individual is going to consume or to correct a high blood sugar. The insulin deliver is calculated based on settings that are determined by a health care professional, Medtronic explains.
Medtronic determined that the vent membrane, a component in the recalled infusion sets, could be blocked by fluid during the process of priming/fill-tubing. An infusion set delivers insulin from the pump to the body. Too much insulin may cause hypoglycemia.
Diabetes presents serious long-term health risks if the blood sugar is not properly controlled. People with diabetes can face complications with the heart and circulatory system, kidneys, eyes and the nervous system.
The attorneys at Parker Waichman have researched injuries caused by many types of defective medical devices and are available to answer questions from individuals who are considering filing a lawsuit.
Infusion sets that have become available since April 2017 include a design update of the faulty component. Medtronic believes the new component reduces the risk of insulin over-delivery after an infusion set change. Medtronic will work with patients to ensure that recalled infusion sets are returned and replaced with new sets that have the updated component. The replacement will be done at no cost to the patient. Medtronic has contacted the U.S. Food and Drug Administration (FDA), along with other regulatory bodies around the world, to share information about the infusion set issue.
Dr. Francine Kaufman, the chief medical officer of the Diabetes Group at Medtronic, said Medtronic is “committed to replacing recalled infusion sets for all patients,” and will work as quickly as possible to complete all exchanges.
Hypoglycemia is the abnormally low level of blood sugar (glucose). Glucose is the body’s main energy source.
Hypoglycemia is often associated with treatment for diabetes, although low blood sugar can occur in people without diabetes, according to the Mayo Clinic. Hypoglycemia, like fever, is not in itself a disease but is an indicator of a health problem. Symptoms of hypoglycemia include:
Without treatment to raise the blood sugar, the individual will develop more serious symptoms including:
People with severe hypoglycemia may appear intoxicated. They may slur their words and move clumsily. The person can be in serious danger if the hypoglycemia goes unrecognized and they don’t receive prompt treatment.
Treatment of hypoglycemia involves steps to quickly bring blood sugar back into the normal range — about 70 to 110 milligrams per deciliter. This can be done by consuming fruit juice or a sugar-sweetened soft drink, or by eating hard candy or taking glucose tablets. A person with diabetes who is experiencing hypoglycemia should seek immediate medical attention if the hypoglycemia does respond to such measures. In the long term, it is important to identify the underlying cause of hypoglycemia in order to avoid future episodes.
Medtronic advised infusion-set customers to use only infusion sets made with the new membrane, starting with their next set change. Medtronic reminds patients that they should carefully follow the steps in the recall notification letter when doing the priming/fill-tubing process. This is especially important if the person only has recalled infusion sets available.
Customers in the U.S. can determine if they have recalled infusions sets by visiting the Medtronic website. Customers outside the U.S. will receive instructions specific to their country.
The U.S. Food and Drug Administration (FDA) asks customers who have experienced issues with the use of a Medtronic infusion set, to report the issue to the FDA’s MedWatch Adverse Event Reporting program. Reporting can be done online, by phone, or by fax. These voluntary reports are important in helping the FDA identify problems with medical devices.
If you or a person someone you know with diabetes has experienced hypoglycemia because of a faulty Medtronic infusion set, you may have cause for legal action. Contact the attorneys at Parker Waichman LLP for a free, no-obligation evaluation of your legal rights. To reach the firm, fill out the online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


