
Bayer’s permanent birth control device off the market Congressman Mike Fitzpatrick will be introducing legislation seeking to remove premarket approval. The U.S. Food and Drug Administration (FDA) is facing pressure from members of Congress who want Essure, Bayer’s permanent birth control device, off the market. ABC News reports that Congressman Mike Fitzpatrick will be introducing […]
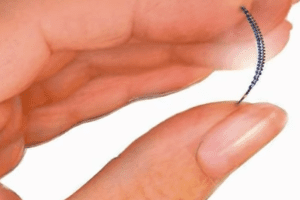
Congressman Mike Fitzpatrick will be introducing legislation seeking to remove premarket approval. The U.S. Food and Drug Administration (FDA) is facing pressure from members of Congress who want Essure, Bayer’s permanent birth control device, off the market. ABC News reports that Congressman Mike Fitzpatrick will be introducing legislation seeking to remove premarket approval. The FDA approved Essure in 2002. Bayer marketed it as a nonsurgical option for women seeking permanent birth control, but it has come under increased scrutiny in light of complaints.
The device consists of two springs inserted into the fallopian tubes to prevent pregnancy. ABC 8News reported that the FDA received over 5,000 complaints related to the device. In light of safety concerns, the FDA held an advisory panel to discuss Essure and make recommendations for its use. The panel advised limited use, and the FDA has not yet made a decision.
The legislation, if passed, seeks to remove the approval within 60 days. The FDA is also facing pressure from another Congress member; Connecticut Congresswoman Rosa Delauro said she was “deeply concerned about the severe adverse health effects of Essure” in a letter to the acting FDA Commissioner. “Essure’s benefits do not outweigh the risk and it should be withdrawn from the market.” she writes.
The personal injury attorneys at Parker Waichman offer free, no-obligation case evaluations. For more information, fill out our online form or call 1-800-YOURLAWYER (1-800-968-7529).
Our essure birth control side effects attorney is here to help you when you need it the most.


