
Inferior Vena Cava Filter Lawsuit Lawyers Provide an Update on Inferior Vena Cava Filter Litigation Inferior vena cava filters are medical devices that surgeons implant into patients who may be at high risk of blood clots and deep vein thrombosis. Blood clots form when the blood does not flow normally or when there is a […]
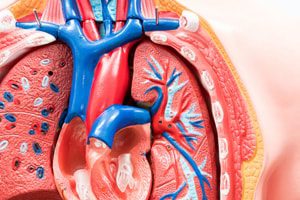
Inferior vena cava filters are medical devices that surgeons implant into patients who may be at high risk of blood clots and deep vein thrombosis.
Blood clots form when the blood does not flow normally or when there is a buildup of plaque in the arteries. Deep vein thrombosis may develop when a blood clot forms in a vein located deep within the body, often in the lower leg or thigh. If left untreated, blood clots and deep vein thrombosis may lead to severe complications or death.
Certain medications may help prevent blood clots or deep vein thrombosis. These medications are called anticoagulation drugs. However, not all patients can take anticoagulants. Other patients who have tried anticoagulants find that they are not as productive as they had hoped. For these patients, an inferior vena cava filter was thought necessary. These devices peaked in popularity several years ago; in 2010, it is estimated that 120,000 inferior vena cava filters were implanted in patients throughout the United States. However, after lawsuits began popping up and the FDA began issuing warnings on the use of IVC filters, surgeons were more hesitant to implant them. By 2014, around 96,000 were surgically implanted in American patients, a significant drop from previous years.
Inferior vena cava filters, or IVC filters for short, are supposedly designed to catch blood clots that travel through the vena cava—like a trap. The vena cava is a principal vein that transports blood to the heart. However, critics argue that there is little scientific evidence to support these claims. The United States Food and Drug Administration has recommended that as soon as the threat of blood clots passes, IVC filters should be removed.
Cook IVC Filters
Bard IVC Filters
Cordis Filters
B. Braun Filters
Argon Filters
Other models may also be on the market.
Thousands of individuals have alleged that the IVC filters that were implanted into their bodies have caused injuries—and in some cases, even death. There are thousands of claims pending against Cook and Bard alone.
These lawsuits, as well as the claims that are filed against other IVC filter manufacturers, allege that the devices caused severe complications, such as punctured veins or organ damage. Some suits claim that the IVC filters migrated to other areas of the body or that they are too delicate and may break down over time.
Many IVC filter manufacturers have used an FDA loophole to obtain approval for the devices. According to the 510(k) rule embedded within FDA regulations, medical device manufacturers may receive FDA approval so long as a new product is similar to a product that is already on the market. Therefore, these manufacturers were able to sell IVC filters to physicians and patients, even though the devices had not gone through any rigorous testing or clinical trials.
In one claim, C.R. Bard was ordered by a jury to pay injured patients nearly $4 million after they deliberated at trial. The trial concluded just last month in Florida. It is predicted that the verdict will have significant impacts on subsequent litigation.
Every year, thousands of Americans are injured when their medical devices fail. Thanks to products liability laws, these individuals may be able to file a claim and seek compensation for the harm they have suffered. However, there are specific factors that must be proven for a claimant to succeed in a products liability claim.
Each of these elements must be proven in a products liability claim. If one of these elements cannot be proven, the claim may fail—and the claimant may be solely responsible for the financial harm he has suffered.
Design defects occur when a product’s “blueprints” are flawed. For example, if a cell phone is manufactured with a glass screen that shatters easily, causing injuries to users, the defective nature of the product is that it was designed with that specific type of glass.
Manufacturing defects occur when a problem arises during the production or distribution of a product. For example, if a truckload of food is left in a hot environment for too long, and that food is contaminated and causes illness in consumers, this would be an example of a manufacturing defect.
Marketing defects refer to a lack of proper instruction or warnings with a product. For example, if a medical device should not be implanted in pregnant women because of the risk of injury to the baby, this information should be provided to both consumers and physicians so that pregnant women may seek an alternative treatment. Otherwise, if the pregnant woman or her baby is injured, she may be able to file a products liability lawsuit against the device manufacturer for a failure to warn about the dangers of the product.
A product may contain one type of defect, or it may contain all three types of defects.
Depending on the specific circumstances in the claim, the damages available in a products liability lawsuit may reach into the millions.
Damages may include:
Other damages may also be available. Each products liability claim is unique and possesses a variety of facts that impact the damages award.
If you are considering filing a products liability claim, you should act as quickly as possible to ensure your legal rights are protected. There are deadlines, called the statute of limitations, that limit how long claimants have to take legal action against medical device manufacturers. If a claimant misses these deadlines, the claimant may be forever barred from filing a products liability claim. Therefore, if you think that a medical device has caused your injuries, you should consult an experienced medical device lawyer as soon as possible to discuss your claim.
The Inferior Vena Cava Filter lawsuit attorneys at Parker Waichman LLP aggressively pursue all avenues of recovery to ensure our clients obtain the money they deserve. To schedule your free Inferior Vena Cava Filter lawsuit consultation with our experienced team, call 1-800-YOURLAWYER (1-800-968-7529) today.


