
UNITED STATES – As reported in an online news article published by www.medicaldesignandoutsourcing.com, Centurion Medical Products has issued a recall of endotracheal connector kits following a recall of more than 6 million Teleflex endotracheal tubes and connectors. Centurion Medical Products manufactures connector kits that contain Teleflex endotracheal tubes and connectors, totaling more than 6 million, […]
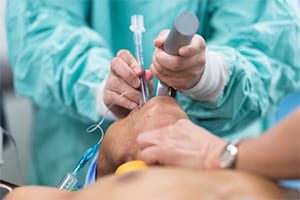
Endo connector kits recall
UNITED STATES – As reported in an online news article published by www.medicaldesignandoutsourcing.com, Centurion Medical Products has issued a recall of endotracheal connector kits following a recall of more than 6 million Teleflex endotracheal tubes and connectors.
Centurion Medical Products manufactures connector kits that contain Teleflex endotracheal tubes and connectors, totaling more than 6 million, that are currently subject to recall. Centurion has issued a recall of 80 of its connector kits.
The United States Food and Drug Administration (FDA) has designated the recall as Class I, the most serious designation possible. Class I recalls involve medical devices that pose a risk of serious injury or death to patients.
Teleflex’s endotracheal tubes and connectors are used on patients under general anesthesia to administer oxygen. Telex initiated numerous recalls of its Hudson RCI Sheridan and Sheridan endotracheal tubes in June 2019 following reports of serious injuries and death.
Teleflex received complaints that the Sheridan connector dislodged from the endotracheal tube and disconnected patients from the breathing circuit which has the potential to result in insufficient oxygenation, decreased vital signs, and death.
When Teleflex issued its first recall, the company was aware of 2 deaths linked to Sheridan endotracheal tubes and connectors. Since the first recall, 2 additional patients have died, and there have been 18 reported injuries. Centurion has not received any reports of injuries or death regarding its connector kits that contain the Teleflex tubes and connectors.
The Centurion connector kits subject to recall have model number TC7855 and were distributed between November 28, 2018, and June 5, 2019. Centurion sent a customer notification letter to Medline, its parent company (and sole customer of the connector kits), instructing Medline to notify all affected customers of the recall with instructions to return the kits.


