
Sorin 3T Heater-Cooler Infected Some Patients with Bacteria, Lawsuit Alleges. According to News Inferno, class action litigation has been filed against medical device manufacturer LivaNova. LivaNova manufactures a product called the Sorin 3T Heater-Cooler system. The lawsuit claims that the device caused bacterial infections in some patients undergoing heart surgeries. The Sorin 3T Heater-Cooler assists […]
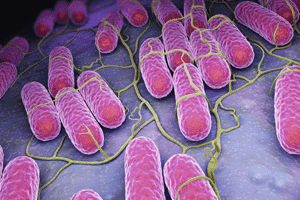
Sorin 3T Heater-Cooler Infected Some Patients with Bacteria, Lawsuit Alleges. According to News Inferno, class action litigation has been filed against medical device manufacturer LivaNova.
LivaNova manufactures a product called the Sorin 3T Heater-Cooler system. The lawsuit claims that the device caused bacterial infections in some patients undergoing heart surgeries. The Sorin 3T Heater-Cooler assists with maintaining the temperature of the patient’s blood and organs. To maintain the temperature, the Sorin 3T Heater-Cooler uses a water tank. The water’s temperature is regulated, and the water itself never makes contact with the patient. However, the United States Food and Drug Administration has warned that contaminated water could become aerosolized, launching contaminants into the air.
The FDA released a Safety Communication in the fall of 2015. The FDA warned about the link between heater-cooler devices and nontuberculous mycobacterium infections. The FDA’s warning read, “Although the water in the circuits does not come into direct contact with the patient, there is the potential for contaminated water to enter other parts of the device or transmit bacteria through the air (aerosolize) through the device’s exhaust vent into the environment and to the patient.”
From January of 2010 to August of 2015, the FDA said that 32 infections had been reportedly caused by these devices. Just in 2015, 25 reports were recorded.
In healthy individuals with strong immune systems, nontuberculous mycobacteria are not usually a threat. These bacteria are generally found in soil and water. However, in patients who have compromised immune systems, or in those who are already sick, the bacteria can cause serious complications.
In June of 2016, the FDA published a subsequent Safety Communication. The warning targeted the Stockert 3T Heater-Cooler System. This device is manufactured by Sorin Group Deustchland GmbH, which now operates as LivaNova.
The release stated a study out of Europe concluded the use of Sorin 3T Heater-Coolers is directly linked to a risk of infections caused by Mycobacterium chimaera. Mycobacterium chimaera is a species of nontuberculous mycobacteria.
The Safety Communication noted,
“The results of this paper suggest a direct link between the M. chimaera to which the European patients were exposed and became infected during open-chest cardiac surgery, and one specific heater-cooler model—the 3T.”
Four months later, in October, the FDA published another release. The release said that the Stockert 3T Heater-Cooler is linked to M. chimaera infections in specific locations within the United States. The FDA added that whole genome sequencing was conducted on water used in the 3T, in air samples taken during the use of the 3T, and from the patients themselves. The FDA stated, “Whole genome sequencing was completed on clinical isolates taken from infected patients and samples taken from 3T heater-cooler devices in hospitals where clusters of patient infections were identified. Samples of the water drained from the 3T devices and air samples collected while the devices were in operations were tested. The results strongly suggest the 3T heater-cooler units are the source of M. chimaera contamination.”
The present class action lawsuit accuses LivaNova of failing to disclose the risk of infection.
Manufacturers, including those that create medical devices, may be liable if their products cause injuries to consumers. Those injured by these devices may seek compensation in a products liability lawsuit.
A products liability lawsuit generally alleges at least one of the following:
In a single products liability lawsuit, one, two, or all three of these allegations may be raised against a manufacturer.
If defective manufacturing is alleged, some type of defect occurred between the product’s creation in a factory and the patient’s receipt of the device. For example, perhaps the device was a type of knee replacement that was created with the wrong material, rendering the device too weak to last more than a year or two once implanted.
If the product was defectively designed, there are no issues with the manufacturing process, but in the inherent design of the product itself. In the knee replacement example, if the device had been originally designed with the weaker material, this is an example of a design defect. Since the product was designed in such a way that made it likely to break and cause injury to patients, it is a design defect.
Defective marketing typically refers to a lack of proper warnings or instructions produced with a product. For example, if a drug is known to cause birth defects in pregnant women, and the information pamphlet with the medication or its labeling does not mention the risk, the company may be liable for damages.
In a medical device products liability claim, there are a number of defendants that may potentially face liability for a victim’s injuries. These defendants include:
Any of these defendants could face liability in a medical device claim. With the assistance of an experienced products liability lawyer, all of the potential defendants will be properly identified in your case.
In a lawsuit, damages are awarded to compensate a plaintiff for the harm or he she has suffered due to the defendants’ negligence. In a medical device products liability claim, damages may include:
Other damages may also be available, depending on the facts of the case. Your attorney will carefully study the evidence in your case, how your injuries have impacted you, and will use this information to build a strong damages claim on your behalf.
If a medical device has caused your injuries, you should meet with an experienced medical device injury attorney as soon as possible. There are deadlines in place that limit how long you have to file products liability claims, so it is important to meet with an attorney to ensure all applicable deadlines are met. If these deadlines are missed, you may forever lose the right to file a lawsuit.
At Parker Waichman LLP, our injury attorneys are experienced in medical device claims. Our attorneys aggressively pursue all avenues of recovery to ensure you obtain the compensation you deserve. To schedule your free consultation, call 1-800-YOURLAWYER (1-800-968-7529).


