
On September 24, 2015, the Obstetrics and Gynecology Devices Panel of the Medical Devices Advisory Committee of the Food and Drug Administration (FDA) will meet to discuss the risks and benefits of Bayer HealthCare’s Essure birth control system. The panel will seek expert scientific and clinical opinion on the risks and benefits of the Essure, […]
FDA Convening Panel to Reconsider Safety of Essure
On September 24, 2015, the Obstetrics and Gynecology Devices Panel of the Medical Devices Advisory Committee of the Food and Drug Administration (FDA) will meet to discuss the risks and benefits of Bayer HealthCare’s Essure birth control system.
The panel will seek expert scientific and clinical opinion on the risks and benefits of the Essure, a method for nonsurgical permanent female sterilization, and will hear testimony from doctors and scientists, as well as from women who have had the device implanted. The committee will evaluate scientific evidence about the safety and effectiveness of the Essure System, including perforation/migration of the device, device removal, chronic pain, allergic reactions, unintended pregnancy, and ectopic pregnancy. The committee will provide recommendations to the FDA about appropriate use of the device, product labeling, and the potential need for additional postmarket clinical studies.
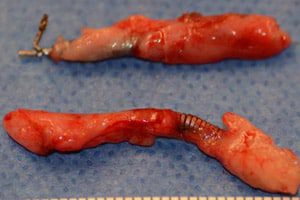
The FDA granted approval for Essure in November 2002. According to Bayer HealthCare Pharmaceuticals’ online materials, the implant procedure can be done in a doctor’s office, takes about 10 minutes to perform, and does not require hospitalization, incisions, or the recovery time of surgical sterilization. Small nickel-titanium coils are inserted through the vagina into the woman’s fallopian tubes. The coils cause scar tissue to form in the fallopian tubes, eventually blocking the tubes and preventing pregnancy. Three months after the procedure, the woman returns for a follow-up X-ray using dye to confirm that the tubes are fully blocked. Once blockage is confirmed, the method is considered 99.83 percent effective, National Public Radio (NPR) reports.
Many women who have received the Essure implant have reported injuries and side effects they say were caused by Essure. These include unintended pregnancies and ectopic pregnancies. Ectopic pregnancy, where the embryo implants somewhere other than the uterus, can be life threatening. A recent NPR report recounts the case of a woman who had Essure implanted in September 2010. She told NPR she experienced chronic fatigue, migraines, joint pain, digestive issues, back pain, heavy periods full of clots, difficulty concentrating, abdominal pain, hair loss, tooth deterioration, depression, and severe bloating, and eventually she had surgery to remove the device. A Facebook group drew posts from more than 18,000 women who have experienced symptoms they attributed to Essure.
The panel meeting will be held at the FDA’s campus in Silver Spring, Maryland and will be open to the public. The FDA intends to make background material available to the public no later than two business days before the meeting. Interested persons may present information and opinions to the panel orally or in writing. Written submissions may be made on or before September 4, 2015. Anyone who wishes to make an oral presentation should submit a brief statement of the general nature of the presentation on or before August 24, 2015; contact Shanika.Craig@fda.hhs.gov. If the number of people registering to speak is greater than can be accommodated during the session, the FDA may conduct a lottery to determine the speakers. The contact person will notify interested persons regarding their request to speak by August 28, 2015. Electronic comments may be submitted to Regulations.gov. Written comments should be sent to the Division of Dockets Management, Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852.


