
As injury and death reports associated with the use of robotics, such as the da Vinci robotic surgical system, in the operating room mount, information is beginning to emerge that has prompted federal regulators to take a closer look at the controversial technology. In one case, a woman undergoing a hysterectomy last year was slapped […]
 As injury and death reports associated with the use of robotics, such as the da Vinci robotic surgical system, in the operating room mount, information is beginning to emerge that has prompted federal regulators to take a closer look at the controversial technology.
As injury and death reports associated with the use of robotics, such as the da Vinci robotic surgical system, in the operating room mount, information is beginning to emerge that has prompted federal regulators to take a closer look at the controversial technology.
In one case, a woman undergoing a hysterectomy last year was slapped in the face by the da Vinci surgical robotic device while she was under general anesthesia, according to a HealthTechZone report. The device’s robotic arm loosened and struck the woman in her face. In that case, the surgeon stopped the procedure and continued the procedure using more traditional surgery and the hospital filed a report with the U.S. Food and Drug Administration (FDA).
That report is just one of 500 filed with the FDA over the da Vinci, according to HealthTechZone, citing the Associated Press (AP). The 500 reports have been received from January 2, 2012 and prompted the federal regulator to open an investigation of the devices last month.
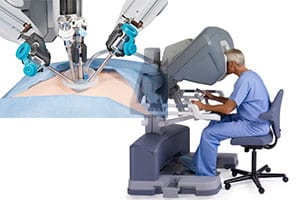
In robotic-assisted surgery, a surgeon sits at a console operating three or four robotic arms, which manipulate small tools that are inserted into the patient’s body by way of tiny incisions. A small, lighted camera displays the surgical area in 3-D video.
The da Vinci is, noted the AP, the only robotic surgery approved by the FDA for soft tissue procedures such as prostate and gallbladder removal, hysterectomies, heart repair, stomach reduction, and organ transplantation. Similar devices have been approved for neurosurgery and orthopedics.
The FDA is now seeking information from surgeons at key hospitals regarding complications seen with the da Vinci and the surgeries for which robotic surgery devices are the best and least suited. Meanwhile, the state-of-the-art system has been the focus of an increasing number of lawsuits and death and injury reports and is at the center of a growing dispute over how the technology is marketed and used.
Prior research published in the Journal of Healthcare Quality reviewed United States hospital websites to better understand how hospital were presenting surgery conducted with robotic devices. Nationwide, 41 percent of hospitals touted procedures conducted with the da Vinci, which is marketed by Intuitive Surgical, according to the HealthTechZone report. Of these, 89 percent claimed that the robotic systems offered clinically superior results compared to traditional surgery, some even using compelling and emotional ads to prompt more patients to use the systems in their surgeries. None of the websites surveyed, noted HealthTechZone, mentioned any risks.
Of the article’s authors, Dr. Martin Makary of Johns Hopkins, has pointed out that no research exists that proves that robotic surgery provides superior outcomes over traditional surgery. And, with the systems costing about $2 million each, there are concerns that the intense marketing efforts have more to do with money than patient health, according to HealthTechZone. Another paper co-authored by Makary reports that physicians are not always reporting negative incidents involving the da Vinci.
da Vinci personal injury lawsuits allege the da Vinci has caused severe internal injuries, including burns, tears, and other complications, some of which resulted in death or chronic pain and disability. Lawsuits fault aggressive marketing tactics used by Intuitive to convince hospitals to purchase the expensive surgical robot, and allege that a combination of design flaws inherent in the robot, coupled with poor physician training on the device, have resulted in serious injuries. Some 70 deaths have been linked to robotic surgical systems since 2009.
In a recent regulatory filing, Intuitive said it faces 26 lawsuits from people alleging injuries associated with its da Vinci surgical system; that it has entered into agreements with some plaintiffs’ attorneys to temporarily suspend the statutory deadline for filing suits alleging injury due to da Vinci surgery; and acknowledged it has seen a “substantial increase” these types of claims, said Bloomberg News.


