
Federal health officials have drastically increased the amount of confirmed cases of fungal meningitis linked to the use of a contaminated vial of an epidural steroid drug injection. According to Reuters, the Centers for Disease Control and Prevention this weekend updated the total number of cases confirmed in the outbreak of a non-contagious form of […]
 Federal health officials have drastically increased the amount of confirmed cases of fungal meningitis linked to the use of a contaminated vial of an epidural steroid drug injection.
Federal health officials have drastically increased the amount of confirmed cases of fungal meningitis linked to the use of a contaminated vial of an epidural steroid drug injection.
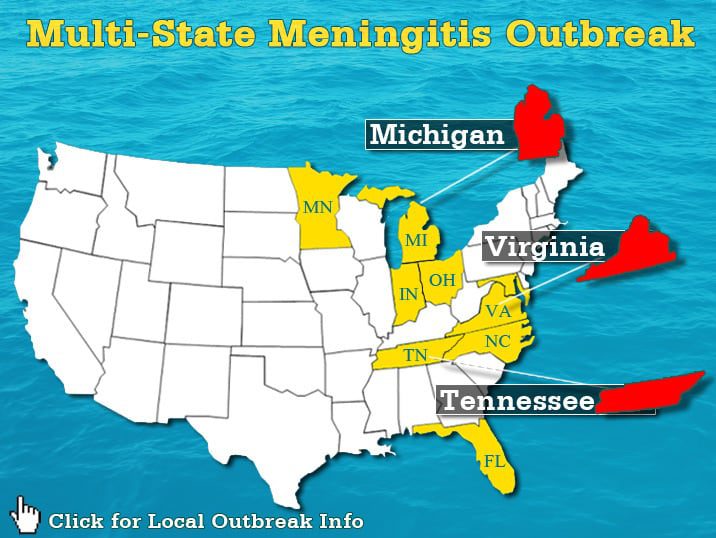
According to Reuters, the Centers for Disease Control and Prevention this weekend updated the total number of cases confirmed in the outbreak of a non-contagious form of fungal meningitis linked to a common painkiller injection that was delivered to 30 states. The agency has now confirmed a total of 205 cases in 14 states. There have been a total of 15 deaths associated with the outbreak.
In the last week, the amount of confirmed victims in this outbreak has doubled. More states have reported victims and CDC officials expect this trend to continue as more recipients of this contaminated injection begin to report their illness and those are confirmed. New cases were recently confirmed in New Hampshire, Florida, Indiana, and Tennessee, according to Reuters.
What has remained constant through the course of the outbreak has been Tennessee reporting the most cases. To date, a total of 53 infections and 6 deaths have been reported in the state. Also, the company at the focus of the outbreak investigation remains the central target and more questions have arisen through the week.
New England Compounding Center processed and distributed vials of methylprednisolone acetate, a painkiller injection that’s delivered in the treatment of back pain and inflammation, from its compounding lab in Framingham, Mass. NECC is one of many pharmaceutical compounding labs across the country. These labs and the compounded or mixed drugs they produce are not regulated by any federal laws or in any way by the Food and Drug Administration. All the drugs and other ingredients they do use in the compounding or mixing processes are supposed to be FDA approved.
NECC, according to previous reports, is only licensed to produce very small amounts of its compounded or mixed drugs. That has raised questions about why and how this company was able to produce enough vials of methylprednisolone acetate to potentially infect 13,000 people in 30 states. The company has issued several recalls in response to the reported fungal meningitis outbreak, including all three Lots of methylprednisolone acetate vials it produced earlier this year. In another recall, the company eventually called back all the products it has produced as a precaution.
It is possible that as many has 5 percent of all recipients of the methylprednisolone acetate could develop a fungal meningitis infection. The number of people, as noted, confirmed in the outbreak is expected to rise. Symptoms of fungal meningitis take up to a month to develop and early symptoms like worsening headaches and fever may not be immediately treated by people suffering them.


