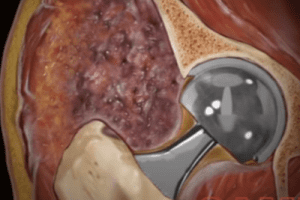
U.K. Claims Against DePuy For Failed Pinnacle Hip Implants
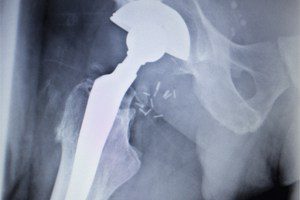
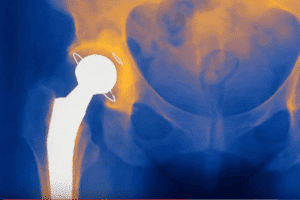
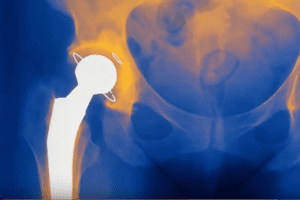
Hundreds of residents of the United Kingdom (U.K.) filed claims against DePuy for defective hip replacement implants. The metal-on-metal implants made by DePuy called the “Pinnacle Ultamet” failed well [...]