
Plaintiffs Allege Merck’s Zostavax is Dangerous. As of mid-July 2017, a total of 18 consumers from nearly one dozen states have initiated a product liability action against Merck & Co Inc. in the Superior Court of New Jersey, County of Middlesex, according to Law360. Allegations include that plaintiffs similarly suffered medical problems after receiving an […]
Plaintiffs Allege Merck’s Zostavax is Dangerous. As of mid-July 2017, a total of 18 consumers from nearly one dozen states have initiated a product liability action against Merck & Co Inc. in the Superior Court of New Jersey, County of Middlesex, according to Law360.
Allegations include that plaintiffs similarly suffered medical problems after receiving an injection with the company’s Zostavax shingles vaccine. Plaintiffs also allege that Merck neglected to warn consumers and medial professionals about the risks associated with the Zostavax vaccine.
Zostavax is intended to prevent shingles; however, the plaintiffs allege that the vaccine caused each of them to contract a persistent strain of the disease. In some cases, this adverse reaction led to postherpetic neuralgia, which is a chronic condition involving nerve damage and pain. One woman who suffered from this reaction said that her symptoms were accompanied by scarring, Law360 wrote.
Zostavax received U.S. Food and Drug Administration (FDA) approval in May 2006. According to the Agency’s approval letter, the Zostavax vaccine is “indicated for the prevention of herpes zoster (shingles) in individuals 60 years of age and older when administered as a single-dose.” Zostavax is a stronger, more potent version of Merck’s chickenpox vaccine, Varivax. According to Law360, Zostavax contains live latent varicella zoster virus whose virulence is reduced or “attenuated,” according to the lawsuit.
The lawsuit indicates that “a risk of using a live virus vaccine is that it is not weakened enough or ‘under-attenuated.’” The lawsuit allegations also pointed out that, “Under-attenuated live virus creates an increased risk of developing the disease the vaccine was to prevent,” according Law360.
The lawsuit was filed on July 11, 2017 in Middlesex County Superior Court. The lawsuit also alleges that Merck, which is based in New Jersey, either knew or should have known that Zostavax could lead to serious side effects, including a viral infection, but that the drug maker did not sufficiently advise plaintiffs and health care providers about these risks. Allegations also include that Merck knew or should have known about a safer alternative to the Zostavax vaccine, according to Law360.
“If the plaintiffs were equipped with the knowledge of the defective condition and potential harms of the Zostavax vaccine, they would not have purchased it and agreed to have it injected into their body,” the lawsuit indicates. The lawsuit also indicates that, “Merck downplayed the serious and dangerous side effects of its product to encourage sales of the product; consequently, Merck placed its profits above its customers’ safety.”
The 18 plaintiffs involved, to date, are from Louisiana, South Carolina, Tennessee, Kentucky, Mississippi, Missouri, Florida, Wisconsin, West Virginia, Michigan, and New Hampshire, according to lawsuit documentation. In these cases, Merck its subsidiary are defendants, as is Ann Redfield, a nurse who worked for the company, and distributor McKesson Corporation, wrote Law360.
Meanwhile, similar litigation brought against Merck over Zostavax is pending in Pennsylvania federal court.
There have been a significant number of reports involving Zostavax that were made to the federal Vaccine Adverse Event Reporting System, according to the lawsuit. As of September 2015, a total of 1,111 submissions were received concerning serious adverse event reports concerning Zostavax, including 36 deaths, according to the lawsuit.
The lawsuit also alleges that the patient information sheet, label, and prescribing information distributed with Zostavax did not appropriately address risks of viral infection and interactions with other common vaccines. Receiving any two such vaccines too closely together can decrease the efficacy of Zostavax, according to the lawsuit. Merck also neglected to alert consumers that the efficacy of Zostavax allegedly decreases with time, according to the lawsuit, wrote Law360. “The Center for Disease Control and Prevention (‘CDC’) published that the Zostavax vaccine wanes in efficacy within five years, having almost no remaining preventative effects after seven years. This allegation is not included on any labeling or packaging literature to alert users of decreased efficacy of the vaccine with time,” according to the lawsuit.
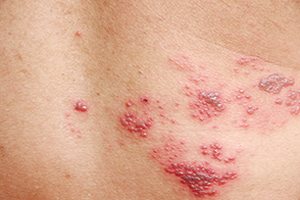 Zostavax is similar to the vaccine use for chickenpox; however Zostavax is about 14 times stronger, which many critics say is far too potent and leads to serious side effects. Merck & Co. claim that the vaccine provides a 50 percent reduction in the likelihood of developing shingles.
Zostavax is similar to the vaccine use for chickenpox; however Zostavax is about 14 times stronger, which many critics say is far too potent and leads to serious side effects. Merck & Co. claim that the vaccine provides a 50 percent reduction in the likelihood of developing shingles.
According to The National Vaccine Information Center (NVIC), Merck’s Zostavax vaccine is the only live virus shingle vaccine on the market. The vaccine contains the live varicella zoster virus, which is the chickenpox virus and which also causes shingles. Once an individual has had the chickenpox, the virus remains dormant in the body. Later, should the immune system become weakened, the virus may re-activate, which lead to shingles.
According to Healthland.com, shingles (zoster or herpes zoster), is a very painful skin rash caused by the virus that causes the chicken pox (varicella zoster) virus. People who have had chicken pox previously, may still develop shingles, which is less contagious than chicken pox and is not passable from person to person. WebMD notes that research from the 1950s revealed that recovery from childhood chickenpox infections does not remove the varicella zoster virus, which remains latent in nerve cells.
The shingles rash typically involves a particular dermatome (a skin area supplied by the involved nerve) that typically appears on one side of the body or face; however, the rash may be widespread. Prior to rash symptoms, patients may experience nerve symptoms such as pain, itching, burning, or tingling. The rash then blisters; the blisters take about one week to scab over. Shingles are not contagious; however, the virus may spread to others and may cause chickenpox.
Approximately one in five people diagnosed with shingles suffer from post-herpetic neuralgia, also known as zoster-related pain that occurs where the shingles rash occurred, even after the rash disappears. The neuralgia may last for several weeks, months, and longer. The older a patient is when diagnosed with zoster, the greater the risk of developing post-herpetic neuralgia.
The personal injury attorneys at Parker Waichman LLP offer free, no-obligation case evaluations. For more information, fill out our online contact form or call 1-800-YOURLAWYER (1-800-968-7529).


